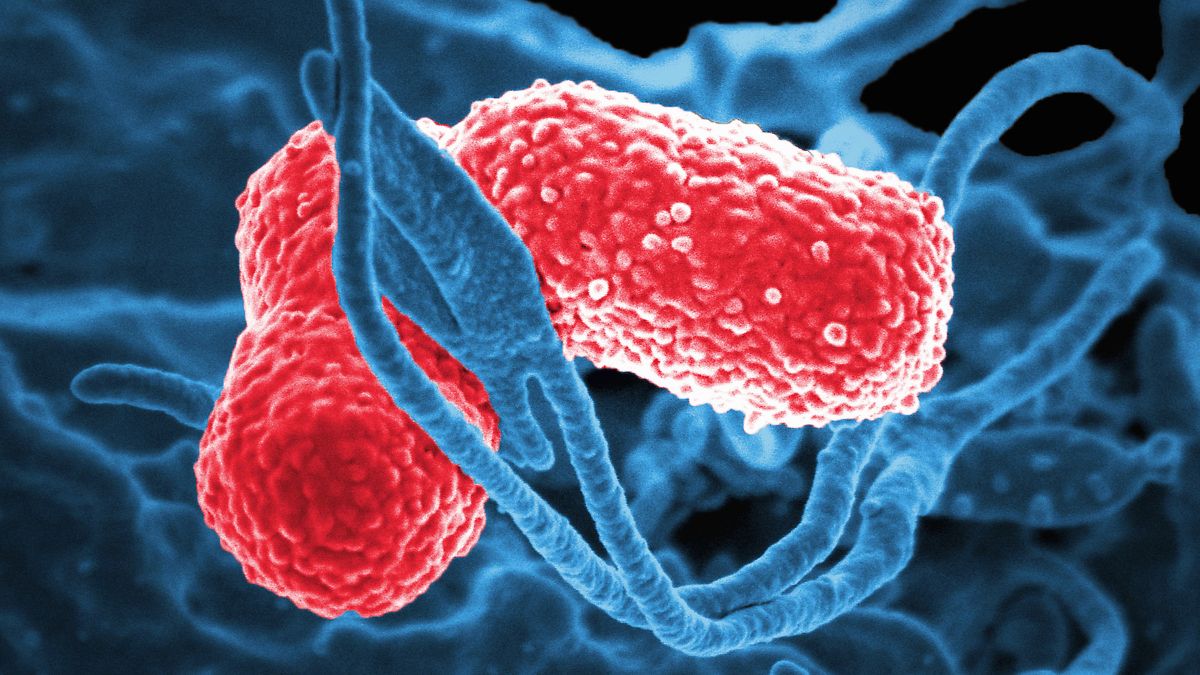
In what is recognized as the most comprehensive investigation of its kind to date, scientific consensus has been reached: bacteria, rather than fungi, are predominantly responsible for auto-brewery syndrome (ABS), a peculiar medical malady causing individuals to experience intoxication subsequent to food consumption without any ingestion of alcoholic beverages.
Through the meticulous analysis of fecal specimens from 22 patients formally diagnosed with ABS, alongside their unaffected household counterparts, investigators discerned two prevalent bacterial species that exhibit significantly higher concentrations in individuals afflicted with the syndrome. This finding corroborates the conclusions drawn from prior research conducted in 2019.
Instances of auto-brewery syndrome are seldom identified, and a definitive therapeutic strategy remains elusive. Consequently, despite the relatively modest cohort size, this study encompasses a considerable number of patients who underwent rigorous ABS evaluation, and whose gut bacteria demonstrated elevated ethanol output when subjected to laboratory cultivation.
“A substantial proportion of affected individuals seek care at multiple medical facilities, only to be erroneously categorized as surreptitious alcohol consumers and depart without an established diagnosis,” the investigative team, spearheaded by infectious disease specialist Elizabeth Hohmann of Massachusetts General Hospital and gastroenterologist Bernd Schnabl from the University of California San Diego, stated in their published findings.
The considerable elevations in endogenous ethanol levels frequently precipitate hepatic damage, in addition to severe social, familial, and legal repercussions.
This seminal study was initiated following an overwhelming influx of inquiries directed at microbiologist Jing Yuan of Beijing’s Capital Institute of Pediatrics, from patients expressing an urgent desire for ABS testing.
It was in 2019 that Yuan, who was not a participant in the present investigation, published research postulating that the bacterium Klebsiella pneumoniae was the likely etiology of ABS, contrary to the prevailing hypothesis that implicated commensal yeast species.
By juxtaposing the gut microbial profiles of ABS patients with those of cohabiting individuals, the current investigation effectively mitigated confounding variables related to environmental and dietary influences known to modulate the gut microbiome.
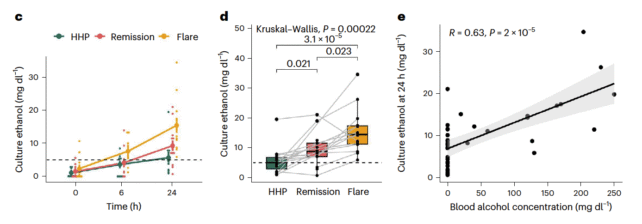
Schnabl and his associates observed that bacterial cultures derived from patients experiencing symptomatic ‘flare-ups’ – manifestations of intoxication – yielded greater quantities of ethanol compared to microbes from individuals in remission or from non-affected household members. This observation showed a direct correlation with measured blood alcohol concentrations during the same temporal intervals.
K. pneumoniae and Escherichia coli, two bacterial genera recognized for their ethanol-producing capabilities, were both found to be more prevalent in ABS patients, with E. coli exhibiting particularly heightened abundance during ABS exacerbations.
Remarkably, one patient experienced a significant amelioration of symptoms following two fecal microbiota transplants from an unaffected donor, which effectively reset his gut microbial ecosystem. The individual remained asymptomatic for over 16 months post the second procedure, with his family reporting that his baseline demeanor had “essentially returned” to normal.
Emerging therapeutic interventions may also be directed at specific bacterial genes implicated in metabolic pathways identified by the researchers as exhibiting increased activity during remission periods, thereby potentially resolving symptomatology.
The research cohort also displayed “extreme” dysbiosis, or imbalances, within their gut microbiomes. Previous investigations have documented diminished ethanol production in individuals with diabetes and have linked gut microbes capable of ethanol synthesis to non-alcoholic fatty liver disease, which stands as the most globally prevalent hepatic disorder.
“This observation prompts a broader inquiry into the prevalence of gut microbial ethanol production within the general populace and the potential extent of its pathological ramifications,” Schnabl and his colleagues articulated.
“Furthermore, our research underscores the critical importance of the gut microbiome and its metabolic byproducts for human well-being,” they concluded.