Historically, β-hydroxybutyrate (BHB) has been recognized as a hepatic substrate serving as an energy source. However, recent scientific investigation has unveiled an additional metabolic role for BHB. A specific enzyme, CNDP2, facilitates the conjugation of BHB with amino acids. Notably, the most prevalent BHB-amino acid derivative, N-β-hydroxybutyryl phenylalanine (BHB-Phe), has demonstrated the capacity to modulate body weight and metabolic processes within experimental animal subjects.
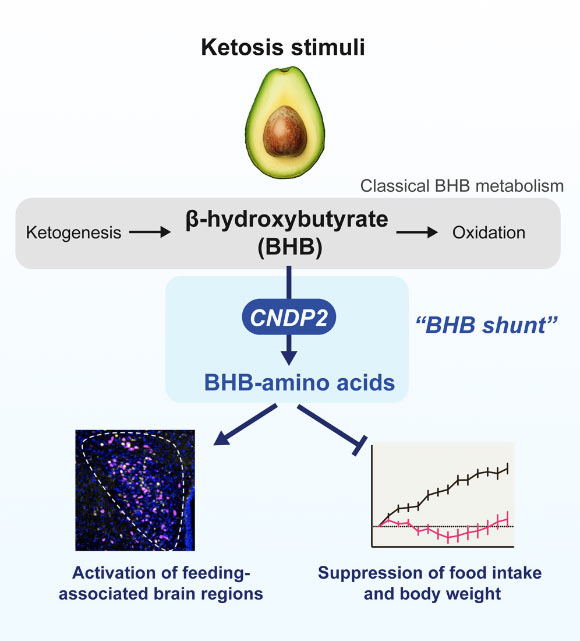
β-Hydroxybutyrate (BHB) is a prevalent ketone body. Previously, all established metabolic pathways for BHB involved the interconversion with primary energy intermediates. Moya-Garzon et al. have identified a novel, previously unknown secondary metabolic pathway for BHB, characterized by the enzymatic conjugation of BHB with free amino acids, dependent on CNDP2. Image attribution: Moya-Garzon et al., doi: 10.1016/j.cell.2024.10.032.
Mammalian organisms have developed sophisticated nutrient-sensing mechanisms that synchronize external energy availability with internal metabolic equilibrium.
These intricate systems involve alterations in cellular energy metabolites, which function both as conduits for energy and as downstream regulatory agents.
A prime illustration is BHB, a ketone body whose concentration escalates during periods of diminished carbohydrate intake, such as during periods of fasting, intermittent caloric restriction, or adherence to a ketogenic dietary regimen.
In this recent investigation, Professor Yong Xu of Baylor College of Medicine, along with his research team, sought to elucidate the impact of BHB-Phe, the most abundant amino acid derivative of BHB, on feeding patterns and body mass in murine models.
Professor Xu stated, “We understood that specific neuronal clusters in the brain govern feeding behaviors, prompting us to undertake a comprehensive brain mapping initiative to pinpoint the regions activated by BHB-Phe.”
“Our findings indicated that BHB-Phe triggers neuronal populations within the hypothalamus and brainstem, thereby inhibiting food consumption and reducing body weight.”
“Conversely, mice engineered to lack the CNDP2 enzyme, and consequently deficient in BHB-Phe, exhibited increased food intake and subsequent weight gain.”
Notably, the CNDP2 enzyme responsible for synthesizing BHB-Phe also generates a structurally similar compound known as Lac-Phe, which this research group had previously characterized.
Professor Xu commented, “Lac-Phe is a circulating compound synthesized during physical exertion that has been shown to decrease caloric intake and combat obesity in mice.”
“The pertinent question arises: do Lac-Phe and BHB-Phe exert their analogous effects through the activation of identical neural pathways in the brain?”
“Our detailed analyses revealed that only a modest subset of neurons responded to activation by both compounds; the majority of neurons influenced by Lac-Phe and BHB-Phe were distinct.”
“This observation suggests the potential for these compounds to modulate feeding behaviors through comparable outcomes but via divergent mechanistic routes.”
The study’s conclusions point towards the possibility that the newly identified BHB-Phe pathway, which is also operative in humans, may be dysregulated in individuals with obesity and potentially other related conditions, underscoring the necessity for further research to thoroughly comprehend its underlying mechanisms.
Dr. Jonathan Long, a research scientist at Stanford University, remarked, “This work unlocks a multitude of novel avenues.”
“For instance, it may become feasible in the future for individuals to ingest BHB-Phe to facilitate weight reduction without the need for carbohydrate restriction in their diet.”
These groundbreaking findings are published this week in the esteemed journal Cell.
_____
Maria Dolores Moya-Garzon et al. A β-hydroxybutyrate shunt pathway generates anti-obesity ketone metabolites. Cell, published online November 12, 2024; doi: 10.1016/j.cell.2024.10.032





