Osteoporosis, a debilitating condition characterized by skeletal fragility, impacts a significant portion of the global population. Promising advancements in therapeutic interventions may be forthcoming following the elucidation of a fundamental mechanism by which physical activity enhances bone density.
This newly uncovered biological pathway presents an avenue for scientists to potentially engineer strategies to counteract the bone weakening associated with osteoporosis. While the positive correlation between exercise and skeletal well-being has been consistently observed, the precise molecular underpinnings have remained largely elusive until now.
“A comprehensive grasp of how our skeletal structures gain resilience through locomotion or exercise is prerequisite to our ability to pharmacologically replicate these benefits at the cellular level,” explains Xu Aimin, a distinguished biomedical scientist affiliated with the University of Hong Kong. “This investigation represents a pivotal progression toward achieving that objective.”
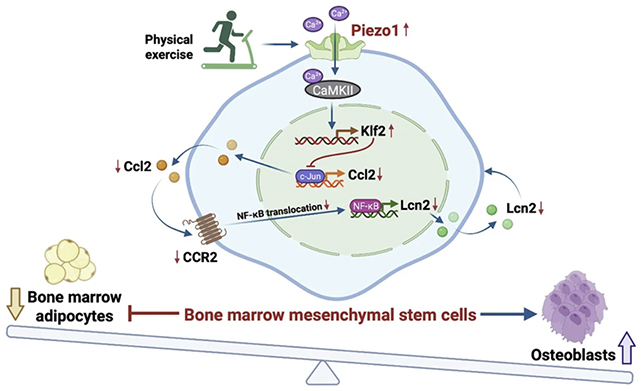
The investigative focus was directed towards mesenchymal stem cells residing within the bone marrow (BMMSCs). These progenitor cells possess the inherent capacity to differentiate into either bone-forming osteoblasts or fat-storing adipocytes.
The developmental trajectory of BMMSCs is influenced by a multifaceted array of stimuli, including growth factors, hormonal signals, inflammatory markers, and – notably in the context of this research – the mechanical forces generated by physical exertion.
Previous investigations utilizing cultured cells had provided indications that mechanical forces favour osteogenesis while inhibiting adipogenesis. However, the underlying molecular mediators remained to be definitively identified. This study pinpointed a protein, Piezo1, known from prior research to transduce mechanical stimuli into intracellular biochemical signals in response to pressure, strain, and stress.
Experimental ablation of Piezo1 in murine models resulted in a noticeable decline in bone mineral density and attenuated bone formation. Concurrently, an observable augmentation in the population of adipocytes within the bone marrow was documented. Furthermore, supplementary analyses revealed that mice deficient in Piezo1 did not experience the exercise-induced enhancements in bone strength.
The researchers subsequently delineated the specific intracellular signaling cascades orchestrated by Piezo1. Their findings revealed that the absence of this protein precipitates inflammatory processes and promotes adipose tissue accumulation. Crucially, these detrimental effects were found to be reversible through either direct activation of Piezo1 or restoration of its downstream signaling. This understanding is paramount for the potential development of future pharmacological agents designed to mimic Piezo1’s function.
“We have effectively deciphered the biological mechanism by which the organism translates physical movement into enhanced skeletal integrity,” states Aimin. “We have successfully identified Piezo1 as the molecular sensor for exercise-induced stimuli and elucidated the signaling pathways it governs.
“This discovery furnishes a precise therapeutic target. By modulating the Piezo1 pathway, we can potentially elicit the salutary effects of exercise, thereby inducing a state analogous to physical activity in the absence of actual movement.”
The natural aging process is often accompanied by a decrease in bone mass, increasing susceptibility to osteoporosis. For many individuals, particularly the elderly and those with compromised physical health, engaging in consistent exercise can be challenging or prohibitive. A therapeutic modality capable of simulating some of the physiological benefits of exercise could offer protection against bone loss in these vulnerable populations.
It is important to note that the translation of these findings into viable clinical treatments remains a distant prospect. This research was conducted using animal models, and interventions targeting a protein with diverse physiological roles like Piezo1 necessitate extreme caution to avoid unintended adverse effects.
Nonetheless, this study, alongside other concurrent investigations, significantly advances our comprehension of osteoporosis pathogenesis. Given the escalating demographic of aging individuals worldwide, there is an urgent imperative to develop strategies that promote sustained health well into later life.
“This presents a potentially transformative approach that extends beyond conventional physical rehabilitation,” observes Eric Honoré, a mechanobiologist and senior author of the study, associated with the Institute of Molecular and Cellular Pharmacology in France.
“In the future, we may be able to deliver the physiological advantages of exercise via targeted pharmacological interventions, thereby mitigating bone demineralization in susceptible groups such as bedridden patients or individuals with restricted mobility, and consequently reducing their fracture risk.”
The findings of this research have been formally published in the peer-reviewed journal Signal Transduction and Targeted Therapy.