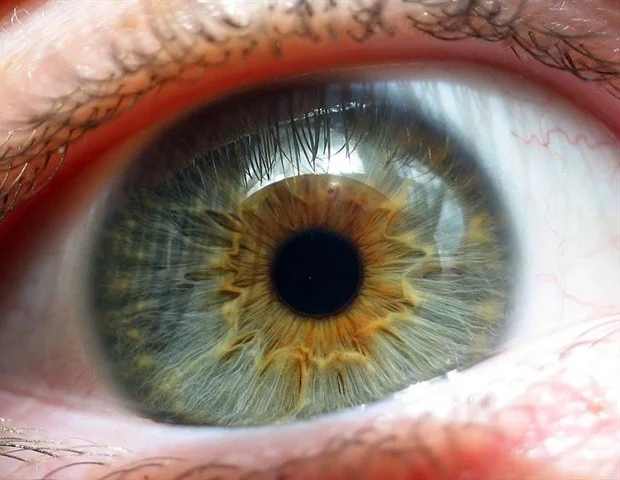
Acquisition of acute visual acuity in prenatal development is facilitated by a synergistic interaction between a vitamin A metabolite and thyroid hormones within the retina, according to researchers at Johns Hopkins University.
These groundbreaking discoveries may necessitate a reevaluation of established paradigms concerning the genesis of light-sensing cellular structures in the eye, potentially steering future investigations toward novel therapeutic strategies for conditions such as macular degeneration, glaucoma, and other age-related visual impairments.
The intricate details of this investigation, which employed laboratory-cultivated retinal tissue, are presently disseminated in the journal Proceedings of the National Academy of Sciences.
“This represents a pivotal advancement in deciphering the internal operational mechanisms of the retinal center, an area of paramount importance within the ocular system and the initial site of deterioration in individuals afflicted with macular degeneration,” stated Robert J. Johnston Jr., an associate professor of biology at Johns Hopkins and the principal investigator of the study. “By achieving a more profound comprehension of this region and developing organoids that effectively emulate its functionality, we aspire to cultivate and implant these tissues in the future to restore sight.”
In recent years, the research contingent has pioneered an innovative methodology for examining ocular development through the utilization of organoids – diminutive clusters of tissue cultivated from embryonic cells. By meticulously observing these artificially grown retinas over extended periods, the scientists elucidated the molecular pathways governing the formation of the foveola, a central retinal zone indispensable for sharp visual perception.
The research specifically scrutinized the light-sensitive photoreceptor cells responsible for diurnal vision. These cells differentiate into blue, green, or red cone cells, each possessing varying sensitivities to distinct wavelengths of light. Despite constituting a mere fraction of the retina, the foveola contributes approximately 50% to overall human visual processing. Intriguingly, the foveola harbors red and green cones exclusively, with blue cones being distributed more broadly throughout the remainder of the retina.
The presence of these three cone types in humans, enabling the perception of an exceptionally broad spectrum of colors, is a distinguishing characteristic that is comparatively uncommon among other species. The developmental trajectory of ocular structures with such a precise cellular distribution has remained an enigma for scientists for decades. As noted by Johnston, model organisms frequently employed in biological research, such as rodents and fish, do not exhibit this cellular arrangement, thereby complicating the study of photoreceptor cells.
The research fraternity at Johns Hopkins has posited that the stratification of cones within the foveola is the outcome of a meticulously coordinated sequence of cell fate determination and transformation during the embryonic stages. Initially, a limited quantity of blue cones is discernible within the foveola between gestational weeks 10 and 12. However, by week 14, these cells undergo a metamorphosis into red and green cones. The study reveals that this intricate patterning is orchestrated by two principal mechanisms: firstly, the enzymatic degradation of retinoic acid, a derivative of vitamin A, which curtails the proliferation of blue cones; and secondly, the influence of thyroid hormones, which actively promote the conversion of blue cones into red and green cones.
Initially, retinoic acid plays a role in establishing the pattern. Following this, thyroid hormone is instrumental in the transformation of the remaining cells. This conversion is highly significant, as the presence of blue cones in this area compromises visual acuity.”
Robert J. Johnston Jr., associate professor of biology, Johns Hopkins University
These findings introduce an alternative perspective to the prevailing hypothesis, which suggested that blue cones actively migrate to other retinal regions during development. Conversely, the accumulated evidence indicates that these cells undergo a transformation to achieve an optimized distribution of cones within the foveola.
“The dominant theoretical framework within the scientific community for approximately the past three decades proposed that the scarce blue cones present in this area simply relocate, assuming these cells are preordained in their identity and remain so permanently,” remarked Johnston. “While we cannot definitively dismiss this possibility, our data strongly advocate for a divergent model where these cells actually undergo a conversion over time, a finding that is quite astonishing.”
These insights hold considerable promise for the development of novel therapeutic interventions for visual impairment. Johnston and his research team are actively engaged in refining their organoid models to more accurately mirror the functional characteristics of the human retina. Such advancements could lead to the generation of enhanced photoreceptors and the potential for cell-based treatments for ocular pathologies like macular degeneration, for which no definitive cure currently exists, according to lead author Katarzyna Hussey, a recent doctoral graduate from Johnston’s laboratory.
“The objective of employing this organoid technology is to ultimately engineer a precisely tailored population of photoreceptors. A significant avenue of potential lies in cell replacement therapy, wherein healthy cells are introduced to reintegrate into the eye and potentially restore lost vision,” explained Hussey, who has since joined CiRC Biosciences in Chicago as a molecular and cell biologist. “These are protracted experimental undertakings, and naturally, extensive optimizations for safety and efficacy studies would be requisite before proceeding to clinical application. Nevertheless, it represents a feasible pathway forward.”
Hussey, K. A., et al. (2026). A cell fate specification and transition mechanism for human foveolar cone subtype patterning. Proceedings of the National Academy of Sciences. DOI: 10.1073/pnas.2510799123. https://www.pnas.org/doi/10.1073/pnas.2510799123