A novel genetic neurodevelopmental disorder, previously unrecognized, has been pinpointed by researchers, heralding the prospect of new diagnostic avenues for countless children and their families globally.
The genesis of this disorder lies in a mutation affecting the RNU2-2 gene. This specific mutation operates recessively, necessitating inheritance from both parental figures to manifest its effects, whereas the gene itself is non-coding, meaning it does not directly synthesize proteins but exerts its influence through indirect cellular mechanisms.
These two distinguishing features—recessive inheritance and non-coding function—provide insight into the prolonged period during which this neurodevelopmental disorder remained undetected. Typically, these areas of our genetic blueprint are not the initial focus when investigating the origins of such health conditions.
An international consortium of scientists engaged in this discovery aimed to scrutinize RNU2-2 with greater detail following the earlier identification of a dominant mutation in the same gene, which had been observed to trigger a severe neurological disorder characterized by pronounced epilepsy. This previously identified condition, however, is less prevalent than the newly identified developmental disorder.
“Our finding offers families a definitive molecular rationale for their child’s condition, something they have often awaited for many years,” observes geneticist and co-lead author Daniel Greene from the Icahn School of Medicine at Mount Sinai, in the United States.
“For numerous families, this newfound clarity can be immensely significant following an extensive and uncertain diagnostic odyssey. Concurrently, it furnishes the scientific community with a tangible biological target to direct future therapeutic interventions.”
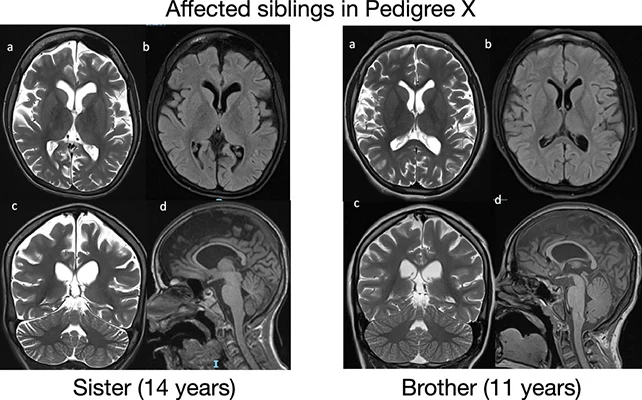
With an estimated prevalence suggesting that ReNU2 syndrome may account for approximately 10 percent of neurodevelopmental disorder cases with a known genetic, recessive origin—potentially affecting thousands of individuals in the UK alone—the researchers indicate that its presentation can include developmental delays, restricted verbal capacity, and diminished muscle tone.
Learning impairments might be apparent, alongside characteristics resembling those seen in autism spectrum disorder and difficulties with ambulation or other forms of motor function. While epilepsy or challenges with respiration and feeding may be involved, the condition appears to manifest uniquely in each affected child.
The identification of ReNU2 syndrome was achieved by the research team through the comprehensive analysis of 110,009 individual genome records sourced from two distinct health research repositories. This involved the selection of 14,805 individuals diagnosed with a neurodevelopmental disorder, who were then compared against a control cohort of 52,861 unrelated individuals without any such disorder.
Subsequently, sophisticated statistical algorithms were employed to pinpoint non-coding genes associated with these conditions and to estimate the potential number of individuals within the general population who might be affected by ReNU2 syndrome. The validity of these findings was further corroborated in selected patients through diagnostic blood analyses.
“This can be regarded as a significant advancement due to the unexpectedly high frequency of these variants—more than triple that of the next most common recessive mutation implicated in severe neurodevelopmental disorders,” states neuroscientist Cornelius Gross of the European Molecular Biology Laboratory in Italy, who was not a participant in this study.
“This discovery holds broad medical importance as it underscores the role of non-coding genes as significant loci for disease development, necessitating increased attention in our ongoing pursuit of the genetic underpinnings of various ailments.”
Beyond providing diagnostic clarity for individuals with the disorder and their families, this investigation is poised to be instrumental in advancing our understanding of how the condition can be managed in the present and potentially prevented in the future.
Nevertheless, considerable obstacles persist. The task of delivering therapeutic agents to the brain and the cells affected by ReNU2 syndrome is anticipated to be challenging. Furthermore, given that the disorder is inherited from birth, the feasibility of retroactive treatment remains uncertain.
“Although a targeted therapy for recessive ReNU2 syndrome is not yet accessible, the understanding that the disorder originates from a deficiency in U2-2 RNA points towards promising gene replacement strategies for future development,” notes geneticist Ernest Turro from the Icahn School of Medicine at Mount Sinai.