Recent scientific findings suggest a particular area of the brain may be implicated in certain instances of elevated blood pressure.
Crucially, this discovery may also unlock a pathway for reversal.
A collaborative effort between researchers at the University of São Paulo in Brazil and the University of Auckland in New Zealand has identified the lateral parafacial (pFL) region of the brain as a potential trigger for physiological adjustments that elevate blood pressure.
The pFL is known for its role in regulating respiration, particularly the robust and intentional exhales associated with physical exertion, coughing, or laughter.
Experimental investigations conducted on rats revealed an additional function: it can induce vasoconstriction.
This dual influence – impacting both respiratory control and vascular signaling – could be a contributing factor to hypertension in some individuals, according to the investigators. This might elucidate why a significant proportion of the population (estimated by some sources to be around 40 percent at a rough estimate) continues to experience unmanaged hypertension despite adhering to antihypertensive pharmacotherapy.
The study posits that pFL neurons might establish a connection between subtle alterations in breathing patterns, which may not be overtly perceptible, and augmented activity within the sympathetic nervous system – the body’s “fight-or-flight” mechanism responsible for managing blood pressure. This aligns with prior research that has established a link between hypertension and the central nervous system.
“Considering that approximately half of all hypertensive patients exhibit a neurogenic component, the primary challenge lies in comprehending the mechanisms responsible for generating sympatho-excitation in hypertension,” the researchers stated in their published findings. They elaborated:
“Such an insight would furnish much-needed clinical guidance for the development of novel therapeutic interventions.”
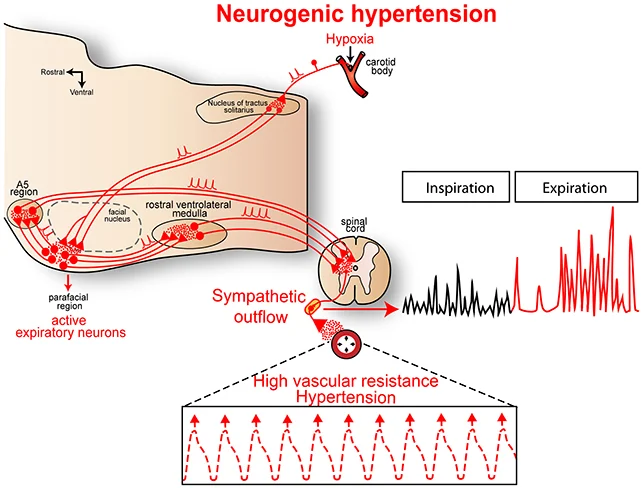
During their experimental protocols involving rats, the research team employed genetic manipulation techniques to selectively activate or deactivate pFL neurons, subsequently observing the resultant physiological effects. Comprehensive monitoring was conducted on respiratory nerve activity, sympathetic nerve output, and blood pressure.
Instances where pFL neurons were stimulated in a subset of the rat subjects led to the activation of other neural pathways that ultimately resulted in elevated blood pressure in these animals.
Subsequent detailed mapping of brainstem and peripheral nerve activity was then performed, including the identification of other neurons with which the pFL region engaged in communication. These findings were contrasted with data obtained from control rats that did not exhibit hypertension.
In rats exhibiting hypertension, pFL neurons not only contributed to respiratory regulation but also actively induced vasoconstriction. This discovery also illuminated a novel potential avenue for therapeutic intervention.
“Our investigation revealed that under conditions of elevated blood pressure, the lateral parafacial region becomes active. When our team successfully de-activated this specific region, blood pressure readings returned to normative ranges,” reported lead physiologist Julian Paton from the University of Auckland.
The information disseminated in this research also offers a partial explanation for the observed correlation between sleep apnea, a condition characterized by nocturnal breathing disturbances, and an increased predisposition to hypertension. This heightened risk has been documented in numerous studies.
The connection between respiratory function and circulatory dynamics is again highlighted: While pFL neurons are not engaged during routine breathing, they are activated in response to elevated carbon dioxide or diminished oxygen levels, physiological states characteristic of sleep apnea episodes.
It is imperative to acknowledge that this research was predominantly conducted using animal models. While it is considered probable, it is not definitively established that the same neural circuitry operates in human physiology.
Nevertheless, given that approximately one-third of the global population is estimated to suffer from hypertension, and a substantial number of these individuals face challenges in accessing effective pharmacological treatments, the imperative for innovative therapeutic solutions is exceptionally pressing. Hypertension significantly escalates the risk of numerous cardiovascular complications and has been associated with a spectrum of other health issues, including cognitive decline.
The subsequent critical step involves determining how pharmaceutical agents can be specifically directed towards pFL neurons without adversely affecting other bodily functions. Encouragingly, the researchers have reported making some progress in addressing this very challenge.
Carotid bodies, which are specialized clusters of cells functioning as minute sensory receptors located in the neck, possess the capacity to influence pFL neuron activity from an extracephalic locus. The investigators theorize that targeting these sensory receptors might be a sufficient strategy to modulate the activity of the pFL region.
“Our objective is to target the carotid bodies. We are in the process of acquiring a new pharmaceutical compound, currently undergoing repurposing by our team, intended to attenuate carotid body activity and remotely deactivate the lateral parafacial region in a safe manner, thereby obviating the need for a drug that penetrates the blood-brain barrier,” stated Paton.
While this therapeutic approach may appear more straightforward than developing medications that cross the blood-brain barrier, it will nonetheless necessitate rigorous and extensive preclinical and clinical evaluation.