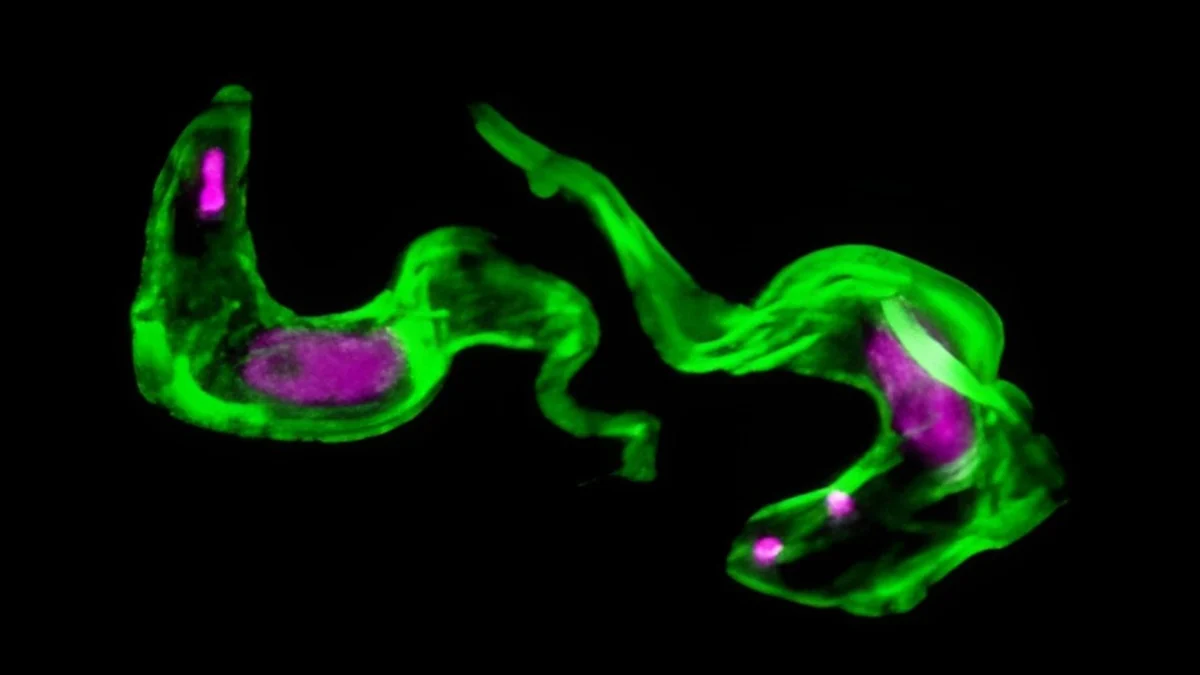
The tropical ailment known as sleeping sickness originates from the African Trypanosoma brucei parasite.
For many years, the precise mechanisms by which this pathogen eludes host immune detection remained elusive, until a recent breakthrough.
It was previously understood that the organism employs a ‘molecular camouflage’ composed of the variant surface glycoprotein (VSG) protein. This external covering undergoes continuous regeneration to circumvent immunological surveillance.
The fundamental enigma centered on the biological machinery facilitating this constant renewal of the protective layer, and specifically, how T. brucei sustains such vigorous activity in this cellular component without compromising the viability of the broader organism.
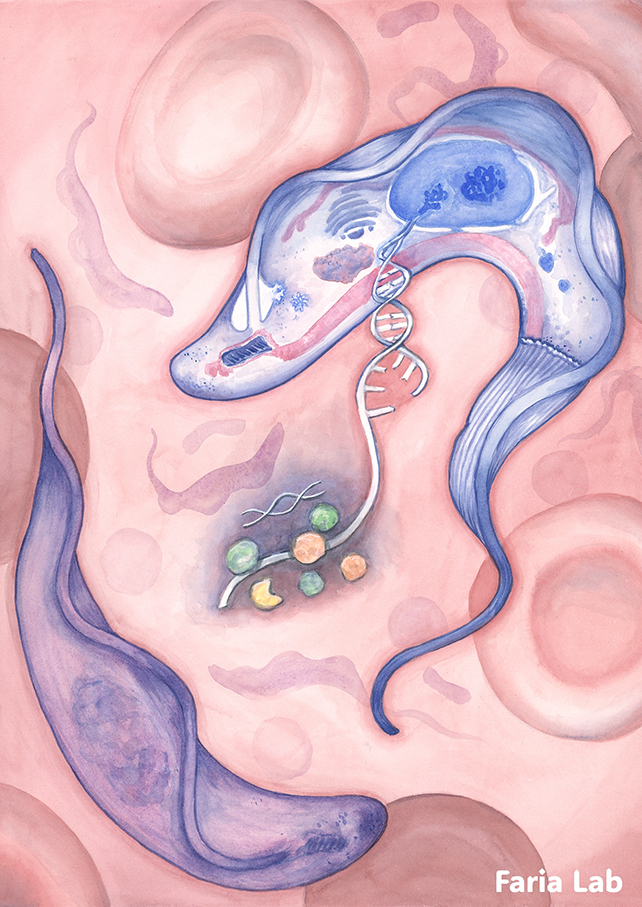
A research consortium from the University of York in the United Kingdom has now elucidated this puzzle, identifying a ‘molecular shredder’ which the investigators have designated ESB2.
This specialized component permits the passage of genetic blueprints for cloak synthesis (in the form of RNA) while simultaneously degrading instructions for other concurrently produced proteins.
“Upon initial microscopic observation of the molecular shredder’s localized presence, we recognized its exceptional significance,” states microbiologist Lianne Lansink, the lead author of a novel publication detailing these findings.
Beyond VSG, the parasite requires auxiliary proteins for its viability, such as those utilized for nutrient acquisition from its host. However, the paramount importance lies with VSG protein synthesis, a process intrinsically linked to ESB2’s function.
Strategically positioned at the terminus of the cellular production pathway, ESB2 exerts precise control over the quantitative regulation of each protein by intercepting specific RNA molecules. This operational strategy ensures sustained, high-velocity VSG production while minimizing energy expenditure.
“Envision a molecular shredder situated within the genetic blueprinting facility, selectively excising particular pages from a manual during its printing process,” elaborates microbiologist Joana Correia Faria, the senior author of the study. “This represents an exceptionally effective method for modulating gene expression at its origin.”
The researchers pinpointed ESB2 through a process involving the labeling of intracellular proteins within the parasite, followed by their identification via mass spectrometry. Subsequent to ESB2’s discovery, the subsequent objective involved ascertaining its functional role. To this end, the researchers employed genetic modification techniques to eliminate ESB2 from T. brucei.
The absence of ESB2 resulted in a dramatic escalation of helper protein production, achieving levels comparable to VSG synthesis. This experimental outcome definitively revealed ESB2’s clandestine function, thereby opening avenues for the development of targeted therapeutic interventions.
“This revelation portends a profound paradigm shift in our comprehension of infectious agents,” comments Faria. “The perpetuation of many organisms may depend less on their methods of disseminating genetic instructions and more on their capacity to neutralize them at their source.”
Sleeping sickness is transmitted via the bite of the tsetse fly and can precipitate a spectrum of neurological disturbances. The pathology can manifest as sleep disturbances, disorientation, and profound comatose states, and is ultimately potentially lethal.
Although the incidence of this disease has seen a decline in recent years, it continues to impact hundreds of individuals annually. Current therapeutic interventions, while available, are not universally accessible, nor do they guarantee efficacy, and can be associated with significant adverse effects.
The groundbreaking research undertaken by the investigators behind this recent discovery offers renewed optimism for the development of novel strategies to combat sleeping sickness.
The immediate future research agenda involves a more granular examination of ESB2, specifically focusing on its mechanisms of recognizing diverse RNA molecules, alongside an investigation into other interacting proteins upon which it may depend.
“This scientific revelation represents a full-circle culmination for me,” shares Faria. “The enduring enigma of how this pathogen orchestrates the differential expression of its genetic instructions has been a persistent, unresolved question since my postdoctoral tenure. It stands as a powerful testament to the novel insights achievable by a contemporary laboratory and a multidisciplinary team of scientists when approaching a long-standing challenge from an entirely fresh perspective.”