A sophisticated three-dimensional “heart-on-a-chip” (HOC) model has been engineered, potentially heralding a significant advancement in combating cardiovascular disease, a condition recognized as the leading global cause of mortality.
A primary obstacle in cardiac research is the inability to safely evaluate drug efficacy or disease progression within a human heart without exposing individuals to considerable risks. This bioengineered cardiac tissue exhibits autonomous contractions, triggers calcium mobilization essential for muscular function, and demonstrates predictable responses to widely used pharmaceuticals.
This innovation distinguishes itself by incorporating a dual-sensing mechanism, enabling continuous monitoring of cardiac activity at the cellular level across the entire tissue construct.
In a recent academic publication, a collaborative team of scientists from various Canadian academic and research centers has detailed their achievement, referring to it as a “substantial leap forward in cardiac tissue engineering and drug evaluation methodologies.”
The pivotal innovation lies in the integration of sensors capable of capturing both macroscopic and microscopic cardiac dynamics. Existing HOC technologies, including the researchers’ prior iteration documented in a 2024 publication, have previously lacked this granular, high-resolution cellular-level sensing capability.
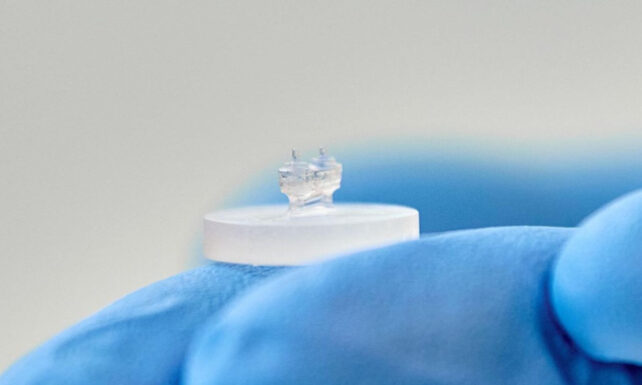
Micro-scale monitoring is of paramount importance, as numerous cardiovascular ailments are intrinsically linked to the compromised function of cardiomyocytes—the individual muscle cells responsible for the heart’s contractile power and the formation of myocardial tissue. Consequently, evaluating the function of these cellular units is crucial for the prophylactic management of heart failure in individuals afflicted with cardiovascular conditions.
To construct their HOC models, the research team isolated cardiac muscle and connective tissue cells from rat subjects. These cells were subsequently cultured within a hydrogel-rich matrix, enriched with structural proteins and essential nutrients to foster cellular proliferation. This engineered tissue was then carefully positioned onto miniaturized, pliable silicon-based substrates.
The researchers strategically incorporated two distinct sensor types into the HOC architecture. For the assessment of gross contractile forces, the engineered cardiac tissues were suspended between two flexible, elastomeric pillars. The degree to which these pillars deform with each cardiac cycle directly correlates with the overall contractile strength of the tissue construct.
Furthermore, the team embedded pliable, hydrogel-based microsensors directly within the cardiac tissue. The subtle deformations experienced by these microscopic structures, which average approximately 50 micrometers in diameter, serve to capture localized mechanical stresses at the cellular level.

This development represents a significant stride towards replicating pathological processes in vitro, given that cellular forces are instrumental in dictating the behavior of cardiac tissues, encompassing their development, structural adaptation, contractile efficiency, tissue repair mechanisms, and even their susceptibility to malignant transformations.
The initial compound administered was norepinephrine, also known as noradrenaline. This sympathomimetic agent, which stimulates the body’s “fight-or-flight” response, is clinically employed to augment cardiac activity and stabilize blood pressure, particularly in critical situations such as cardiac arrest.
To induce the inverse effect and diminish contractile activity, the researchers subsequently applied blebbistatin, a pharmacological agent that inhibits muscle contraction.
The observed drug responses precisely aligned with theoretical predictions, substantiating the HOC’s capacity to accurately forecast the impact of common pharmacologic agents on myocardial force generation and cardiac rhythm.
“The ability to continuously observe the tissue’s reaction to various compounds in real-time offers a substantial advantage for preclinical research and the translational application of findings,” states lead author Ali Mousavi, a biomedical engineer affiliated with the University of Montreal.
Future research endeavors by the scientists involve recapitulating specific cardiac pathologies by cultivating cardiac tissues derived from individuals diagnosed with diverse heart conditions. These conditions include dilated cardiomyopathy, a frequently inherited myocardial ailment that can precipitate heart failure, and various forms of arrhythmias, a collective term for disorders characterized by abnormal heart rhythms.
In the long term, HOC technology holds the potential to empower clinicians to select optimal therapeutic interventions by performing preliminary tests on a patient’s autologous cells prior to prescribing any medication.
“This scientific breakthrough propels us closer to the realization of true precision healthcare,” observes senior author Houman Savoji, a professor of mechanical and biomedical engineering at the University of Montreal, “by equipping us with the capability to identify the most efficacious medication for each individual before any treatment has even been initiated.”
This groundbreaking research has been formally published in the peer-reviewed journal Nano Micro Small.