The cardiac muscle, despite its paramount significance, is one of the body’s tissues that exhibits limited capacity for self-repair following damage – or at least, this has been the prevailing scientific assumption. However, researchers in Australia have recently documented the active proliferation of cardiac myocytes in the aftermath of myocardial infarction.
Instances where blood circulation is compromised result in oxygen deprivation, leading to the demise of cardiomyocytes. While the organ possesses an innate mechanism to mend itself by forming scar tissue, this non-contractile, fibrous connective tissue is incapable of pulsating, thereby diminishing the organ’s overall efficiency. Such functional impairments can ultimately precipitate recurrent cardiac events and culminate in heart failure.
Comparatively, the cardiac systems of rodents appear endowed with a degree of regenerative capability. Their cardiomyocytes (cardiac muscle cells) have been observed to undergo further division subsequent to a heart attack; however, human cardiac cells demonstrate considerably less vitality and proliferative potential post-injury.
“Our investigation reveals that while the heart does develop scarring following a myocardial infarction, it simultaneously generates new muscle cells, which in turn unveils novel therapeutic avenues,” stated Robert Hume, the principal author of the study and a cardiologist affiliated with the University of Sydney.
“While this recent revelation concerning the regrowth of muscle cells is indeed encouraging, it is not yet sufficient to fully counteract the grave repercussions of a heart attack. Consequently, our future objective is to develop interventions that can bolster the heart’s inherent capacity for cell generation and facilitate cardiac regeneration post-event.”
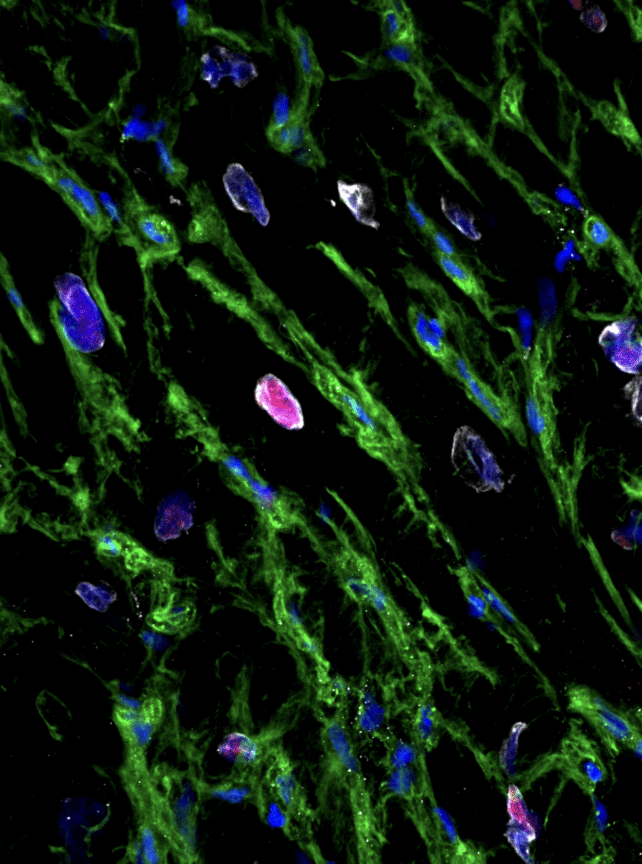
Prior scientific inquiries focusing on patients who had undergone cardiac surgery had previously suggested the latent regenerative potential of cardiac muscle cells subsequent to injury.
In the present investigation, Hume and his collaborators meticulously analyzed living human cardiac tissue obtained from a whole heart donated by a brain-dead individual, alongside samples procured from patients during coronary artery bypass grafting procedures.
The research team conducted comprehensive sequencing of the RNA (which serves as a blueprint for protein synthesis) and performed an in-depth examination of the cellular proteins and metabolic processes within the tissue samples.
The overarching aspiration is that these findings will pave the way for the development of innovative regenerative therapies, potentially offering a future means to address the primary global cause of mortality.