A cardiac arrhythmia known as atrial fibrillation affects millions, characterized by the upper chambers of the heart, the atria, beating in a disorganized and erratic fashion instead of a synchronized rhythm.
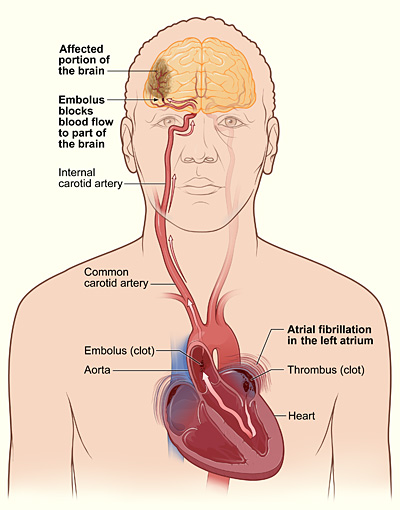
While some individuals experience minor symptoms such as palpitations, fatigue, or dyspnea, the most significant peril associated with this condition is a considerably more grave outcome – a cerebrovascular incident, or stroke.
Within the heart lies a small anatomical feature called the left atrial appendage. When the heart’s rhythm becomes irregular, blood can stagnate in this pouch rather than flowing continuously, and stagnant blood possesses a propensity for clot formation.
Should one of these solidified masses dislodge and travel to the brain, it can obstruct blood flow, thereby precipitating a stroke.
Atrial fibrillation escalates the likelihood of experiencing a stroke by approximately fivefold. Consequently, a paramount inquiry for scientific investigation has been whether this anatomical appendage could be effectively eliminated from contributing to the risk.
Researchers have recently unveiled a potential solution: an innovative methodology, currently evaluated solely in preclinical animal models. This technique involves the introduction of a magnetically guided liquid substance into the heart, which solidifies to permanently occlude the appendage from within.
Initial investigations conducted in rodent and porcine subjects indicate that this therapeutic approach holds promise for mitigating stroke risk in individuals diagnosed with atrial fibrillation in the future.
While existing therapeutic interventions demonstrate efficacy, they are not without limitations. Presently, the mainstay of treatment often involves the prescription of anticoagulant medications. These pharmacological agents impede blood coagulation, thereby substantially reducing the incidence of stroke.
However, the use of anticoagulants is accompanied by inherent risks. They augment the propensity for hemorrhage, posing a significant danger to specific patient cohorts, notably the elderly or those with concurrent medical conditions such as gastrointestinal ulcerations, hypertension, hepatic or renal impairment, and malignancy.
Certain individuals may find these medications intolerable or be compelled to discontinue therapy due to resultant bleeding complications.
An alternative therapeutic strategy is a procedural intervention known as left atrial appendage occlusion. This involves the percutaneous introduction of a device designed to seal the appendage. The most prevalent devices are delivered via catheterization and expand to resemble diminutive metallic umbrellas, effectively occluding the opening.
While these devices can be efficacious, they are not invariably perfect. Given the considerable inter-individual variability in the morphology and dimensions of the left atrial appendage, rigid prostheses may not consistently achieve a hermetic seal.
Consequently, minor blood leakage may occur along the periphery, and the potential exists for diminutive thrombi to precipitate on the device’s surface. Furthermore, the mechanisms by which these devices are secured can inflict iatrogenic damage to cardiac tissues.
The recently elucidated approach represents a paradigm shift. Rather than implanting a rigid prosthesis, investigators administer a magnetically responsive fluid, colloquially termed a magnetofluid, directly into the left atrial appendage utilizing a catheter.
Once introduced into the cardiac cavity, an external magnetic field is employed to guide and anchor the fluid, ensuring its complete inundation of the appendage, even against the pulsatile forces of circulating blood.
Within a brief temporal interval, the fluid interacts with the aqueous components of the blood, undergoing a transformation into a pliable “magnetogel” that effectively seals the chamber.
Owing to its initial liquid state, the substance possesses the capacity to dynamically conform to the intricate and highly variable topography of each patient’s left atrial appendage.
Theoretically, this characteristic facilitates the achievement of a more complete occlusion compared to conventional rigid prostheses.
Additionally, the gel demonstrates an aptitude for integration with the endocardium, establishing a smooth interface that may diminish the likelihood of thrombus formation.
Promising Preliminary Findings
To date, this innovative technique has undergone evaluation exclusively in animal subjects. Researchers initially validated the foundational concept in rodent models before proceeding to more complex experiments involving pigs, a critical juncture in cardiovascular research.
In the porcine study, the magnetogel maintained its structural integrity within the appendage for a duration of ten months, with no observable evidence of thrombosis or leakage.
The endocardial layer of the heart progressively encroached upon the gel’s surface, engendering a continuous and apparently healthy cellular stratum.
When contrasted with conventional metallic occlusion devices in the porcine cohort, the magnetogel facilitated the development of a smoother endocardial lining and obviated the tissue trauma associated with mechanical anchoring elements. Equally significant, the investigators did not detect any adverse biological responses in the animal subjects.
Porcine models are widely adopted in cardiovascular investigations due to the anatomical and physiological parallels between their cardiovascular systems and those of humans, exhibiting comparable dimensions, structural organization, and functional characteristics.
Therefore, the successful demonstration of magnetofluid efficacy and safety within a porcine cardiac environment provides a robust proof-of-concept. However, this does not yet definitively assure the technology’s safety or effectiveness in human subjects.
Despite the encouraging outcomes, the methodology remains firmly situated within the experimental domain. Prior to the initiation of human clinical trials, investigators must substantiate long-term safety, optimize the delivery mechanism for the material, and confirm its predictable behavior in more extensive preclinical animal investigations.
Furthermore, several practical challenges necessitate resolution. For instance, the presence of the magnetic material can interfere with magnetic resonance imaging (MRI) of the heart, potentially compromising visualization of certain cardiac regions.
SuchNotwithstanding these hurdles must be surmounted before clinical implementation. Moreover, the stringent regulatory pathways governing medical devices necessitate protracted testing phases, implying that several years are likely to elapse before its availability for therapeutic application.
Should this technology ultimately prove to be both safe and effective in human trials, it could offer a novel strategy for safeguarding individuals with atrial fibrillation against the debilitating consequences of stroke.
A catheter-delivered liquid occlusion may present a valuable alternative for patients who cannot tolerate anticoagulant pharmacotherapy and could surmount certain limitations inherent in existing occlusion devices.
Considering that atrial fibrillation affects tens of millions of individuals globally, even marginal improvements in stroke prevention could exert a considerable positive influence on public health outcomes.
At present, the magnetically responsive gel represents a laboratory innovation rather than an established clinical therapy. Nevertheless, it underscores the transformative impact of advancements in materials science and biomedical engineering in furnishing novel avenues for addressing one of cardiology’s most enduring challenges.![]()