Groundbreaking scientific inquiry has yielded promising advancements for addressing two of the most significant global challenges we currently confront: the pervasive issue of plastic contamination and the reliance on petrochemicals within pharmaceutical synthesis.
A contingent of researchers from the University of Edinburgh in the United Kingdom has successfully employed strains of Escherichia coli microorganisms to transform constituent molecules derived from the ubiquitous polyethylene terephthalate (PET) plastic into the analgesic compound acetaminophen (also recognized as paracetamol).
A substantial proportion of contemporary pharmaceutical agents, including acetaminophen, are fundamentally synthesized from petrochemical sources. The substitution of these petroleum-derived precursors with discarded materials, such as plastic waste, presents a remarkably innovative strategy to simultaneously mitigate two critical environmental concerns.
While the practical implementation of this methodology on an industrial and commercial scale will necessitate further development and validation, thus warranting a degree of measured optimism, the novel technology exhibits considerable promise.
“This research underscores the potential for PET plastic to transcend its status as mere refuse or a precursor to further plastic production; it can be effectively repurposed by microbial agents into valuable new commodities, including substances with therapeutic applications,” commented Stephen Wallace, a biotechnologist affiliated with the University of Edinburgh, in a recent statement.
The operational sequence commences with the chemical decomposition of PET bottles. Subsequent to this preparatory phase, the resultant molecular fragments are introduced to genetically modified E. coli cultures. These engineered bacteria then utilize phosphate as a catalytic agent to facilitate the conversion of these molecules into a nitrogen-containing organic compound. This intermediate compound is subsequently refined to yield the active pharmaceutical ingredient of acetaminophen.
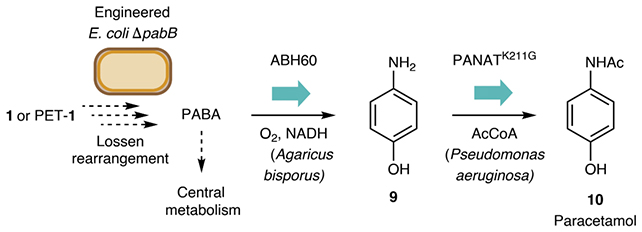
Among the salient advantages characterizing this innovative procedure are its completion within a 24-hour timeframe, its feasibility within a compact laboratory setting, and its operational capacity at ambient temperatures, thereby obviating the requirement for energy-intensive heating or cooling mechanisms. Furthermore, the research team has achieved a remarkably high conversion efficiency, with a yield approximating 92 percent.
The underpinning chemical transformation leverages the well-established Lossen rearrangement reaction, a discovery attributed to the German chemist Wilhelm Lossen in 1872. In this context, the reaction has been rendered biocompatible, enabling its effective function within cellular environments and living bacterial systems.
Although the current demonstration exclusively utilized PET bottles, it is pertinent to note that this type of plastic is also extensively employed in the production of food packaging, furnishings, and a wide array of manufactured goods. It is estimated that this particular plastic constitutes over 350 million tons of annual waste, significantly exacerbating the global plastic pollution crisis.
The researchers suggest that analogous methodologies could potentially be adapted for application with different bacterial species and various plastic types, thereby opening avenues for more environmentally sustainable approaches to recycling and pharmaceutical production.
“The natural world has developed a sophisticated yet finite repertoire of chemical reactions that are fundamental to the functioning of all living organisms,” the researchers expounded in their publication.
“In contrast, the domain of synthetic organic chemistry possesses the capacity to access reactivities not observed in nature. The integration of these abiotic reactions within biological systems offers an elegant resolution to the challenge of sustainably synthesizing numerous industrial chemicals from renewable raw materials.”