Could sharks one day necessitate artificial dental replacements? Emerging scientific findings suggest this may become a reality, as recent investigations indicate that ocean acidification could compromise the formidable bite of sharks.
The dental structure of sharks is a marvel of the natural world, characterized by its exceptional sharpness and continuous renewal. As one tooth detaches, a successor is invariably poised to emerge in its place. This remarkable capacity for dental regeneration is absolutely critical for the survival of sharks, given their evident reliance on their formidable dentition for capturing prey.
However, despite their composition of highly mineralized phosphates and their advanced adaptation for mastication, shark teeth are susceptible to corrosive processes, according to biologist Maximilian Baum of Heinrich Heine University in Germany. This vulnerability is particularly pronounced as ocean acidification intensifies due to anthropogenic carbon emissions.
The world’s oceans serve as a significant carbon sequestration mechanism, absorbing approximately 30 percent of the carbon dioxide (CO2) released into the atmosphere. Nevertheless, as atmospheric CO2 levels ascend, a greater proportion is absorbed by marine environments. This absorption triggers a chemical reaction with seawater, leading to an elevated concentration of hydrogen ions, a subsequent reduction in pH, and ultimately, ocean acidification.
Furthermore, this phenomenon diminishes the availability of carbonate ions, thereby impeding the formation of calcareous structures like seashells and coral. This disruption propagates through aquatic ecosystems, adversely impacting a diverse array of marine organisms, ranging from mollusks and echinoderms to corals and plankton.
Presently, the mean global ocean pH stands at 8.1, a value comparable to baking soda. Projections indicate that by the year 2300, this pH is anticipated to decline to 7.3, rendering the oceans almost ten times more acidic than they are today.
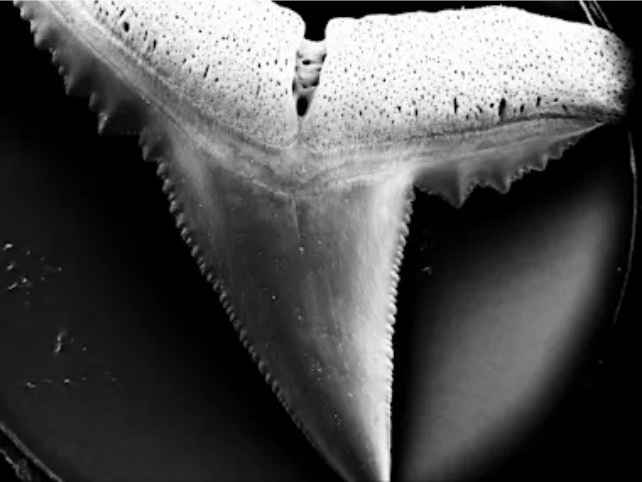
To meticulously evaluate the potential ramifications of future ocean acidification on shark dentition, researchers procured over 600 naturally shed teeth from blacktip reef sharks (Carcharhinus melanopterus) originating from the Sealife Oberhausen aquarium in Germany.
Blacktip reef sharks play an indispensable role within tropical coral reef ecosystems, and their multi-rowed dentition maintains continuous contact with the surrounding seawater. Due to their passive respiratory mechanism, these sharks must undulate forward with their mouths agape to facilitate the passage of oxygenated water across their gills.
Subsequently, the research team meticulously incubated sixteen of the most pristine discarded teeth in separate aquatic environments containing artificial seawater. One environment was maintained at a pH of 8.2, mirroring current oceanic conditions, while the other was adjusted to a pH of 7.3, representative of projected future oceanic environments.

Teeth subjected to incubation in the more acidic aqueous solutions exhibited a statistically significant augmentation in the presence of fissures and perforations. Every constituent of the dental structure experienced detrimental effects: the acidification induced corrosion of the tooth crowns, degradation of the roots, and frequently, an obliteration of the fine distinctions within the serrated edges.
Intriguingly, a marginal increase in the average circumference of the teeth was observed, particularly at elevated pH levels. The authors posit that this phenomenon is indicative of an augmentation in surface irregularities rather than an actual volumetric expansion of the teeth.
While an elevated degree of irregularity could, in principle, enhance cutting efficacy (given that serrations are inherently irregularities), it concurrently renders the teeth more fragile and prone to fracturing.
These findings carry significant implications for a multitude of marine species, including sharks, which are already confronting substantial threats from overfishing.
Ocean acidification may also precipitate a reduction in growth rates and an increase in nutritional demands, which would be exceedingly difficult to satisfy with compromised dentition. Furthermore, other shark species might contend with diminished hatching success or a reduced sensitivity of their chemoreceptors in a more acidified oceanic milieu.
The corrosive effects could also lead to the deterioration of sharks’ dermal denticles, which function analogously to scales, covering their bodies and exhibiting a compositional similarity to teeth. These integumentary structures provide both protective shielding and enhanced hydrodynamic efficiency. As these denticles degrade, sharks could experience a diminished capacity for propulsion, thereby incurring augmented energetic expenditures.
In summation, this research underscores the far-reaching, unanticipated, and complex ramifications of climate change. As Baum eloquently explains, “It serves as a potent reminder that the impacts of climate change reverberate throughout entire food webs and ecosystems.”
The authors, however, do acknowledge certain limitations inherent in their study, notably their utilization of discarded teeth. The precise impact of ocean acidification on teeth still affixed to living sharks may diverge from these observations.
Moreover, the overarching consequence of acidification remains somewhat indeterminate, as certain elasmobranchs (a classification of cartilaginous fish encompassing sharks, skates, and rays) have demonstrated an ability to maintain homeostasis of their blood pH levels amidst shifting environmental conditions. Nevertheless, scientific inquiry will have until the year 2300 to meticulously elucidate these finer details.
This scientific investigation has been formally published in the peer-reviewed journal Frontiers in Marine Science.






