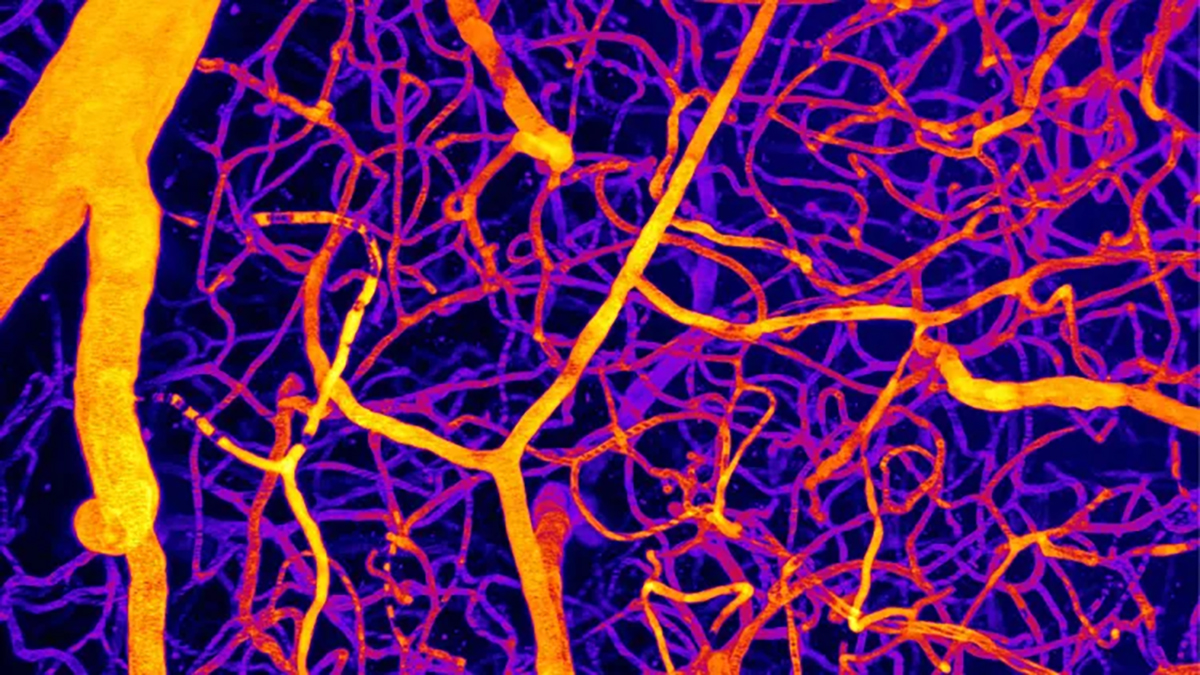
A significant contributor to various forms of cognitive decline, including Alzheimer’s disease, is believed to be compromised cerebral circulation. Recent scientific endeavors have elucidated a novel regulatory pathway governing this vital flow, potentially shedding light on its pathological deviations.
An investigation by the University of Vermont’s research team revealed that a lipid molecule plays a crucial role in preserving the equilibrium of this system. In murine models exhibiting Alzheimer’s pathology, a disturbance in this delicate balance was observed to precipitate adverse effects.
Rectifying this dysregulation yielded a restoration of more normalized blood flow, presenting a promising novel avenue for comprehending and addressing the neuropathological sequelae associated with dementia.
“This groundbreaking research represents a substantial advancement in our pursuit of mitigating dementia and neurovascular disorders,” commented pharmacologist Osama Harraz.
Building upon prior investigations into endothelial cells, which form the inner lining of vasculature, the researchers specifically examined the protein Piezo1, functioning as a mechanosensor within these cells. Aberrant activation of this sensor can lead to disruptions in cerebral blood flow.
Through meticulous analysis of brain activity in laboratory mice, it was ascertained that a lipid, designated PIP2, acts as an inhibitory agent for Piezo1. During periods of heightened neuronal activity, PIP2 concentrations diminish, permitting Piezo1 activation and consequently augmenting blood delivery to areas demanding increased metabolic support.

In instances of Alzheimer’s disease modeled in mice, PIP2 levels were found to be detrimentally low. This deficiency resulted in the overstimulation of Piezo1, leading to an excessive influx of blood into non-essential regions and compromising global circulatory efficiency.
Significantly, the re-establishment of adequate PIP2 levels in these experimental subjects largely normalized the cerebrovascular flow patterns.
While an exhaustive understanding of the intricate workings of this mechanism is still in its nascent stages—this study was of limited duration and confined to murine subjects—it represents an encouraging direction for future research into the fundamental drivers of neurodegenerative conditions.
Vascular dementia, characterized by compromised cerebral blood flow, stands as one of the predominant etiologies of dementia, impacting millions globally. Furthermore, circulatory dysfunctions are implicated in the pathogenesis of Alzheimer’s disease, although the accumulation of pathological protein aggregates is often considered a more prominent factor.
Given that blood flow is instrumental in the delivery of vital oxygen and nutrients to the brain, the implications of this research extend beyond dementia. Maintaining an optimal physiological balance is paramount for sustained cerebral function.
“These findings lay the groundwork for a therapeutic strategy aimed at enhancing cerebral perfusion in conditions where Piezo1 activity is dysregulated, with potential ramifications extending beyond the control of brain blood flow,” articulated the research personnel in their published findings.
Despite incremental progress in our comprehension of dementia, considerable unknowns persist regarding the initiation of these diseases and the predisposition of certain individuals. Even in the context of vascular dementia, the full spectrum of contributing factors to circulatory impairment remains incompletely defined.
Investigations such as this contribute to bridging these knowledge gaps by identifying the molecular components involved.
Subsequent research phases will focus on precisely delineating the interaction between PIP2 and Piezo1. A comprehensive understanding of this molecular interplay will be indispensable for modulating this system and potentially restoring healthy circulatory dynamics, and by extension, cognitive function.
“We are progressively unveiling the intricate molecular pathways underlying these debilitating afflictions, and subsequently, we can commence conceptualizing the translation of this biological knowledge into effective therapeutic interventions,” stated Harraz.