The connection between persistent discomfort and melancholic states has been widely acknowledged for a considerable duration.
Among individuals contending with protracted pain, approximately four in ten manifest overt symptoms of depression. However, the underlying rationale for why a subset of those experiencing chronic pain develop attendant depressive disorders remains a subject of intense inquiry.
Scientists have long grappled with understanding this phenomenon and the neurological processes involved. A comprehensive grasp of this intricate relationship could pave the way for proactive interventions to avert the onset of depression.
Our recent scholarly publication, featured in the esteemed journal Science, posits that the answer to this perplexing question is indeed rooted within the neural architecture of the brain.
To facilitate our investigation, we undertook a meticulous analysis of neuroimaging data derived from 14,462 participants enrolled in the UK Biobank cohort. Our comparative approach involved examining distinct participant groups: individuals who had endured chronic pain for a minimum of seven years without exhibiting depressive symptoms, and those concurrently experiencing chronic pain and developing depressive manifestations.
In the latter cohort, the depressive symptoms were either persistent throughout the entire seven-year observation period or emerged after two or four years of follow-up. This structured timeline enabled a granular examination of depression’s trajectory in relation to chronic pain, leveraging the insights provided by brain imaging technologies.
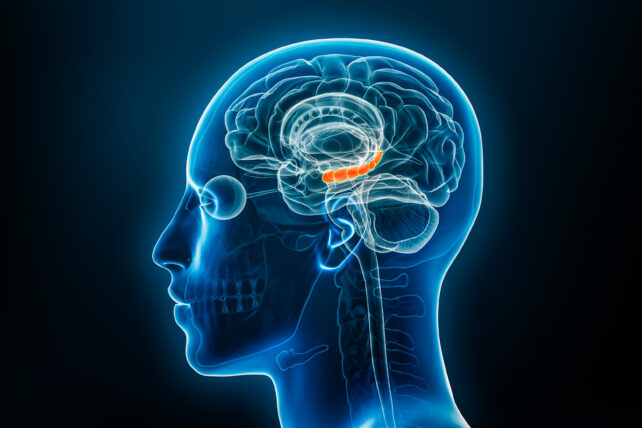
Our analyses of these neuroimaging datasets unearthed a remarkable pattern of activity within a specific brain region: the hippocampus. This area is fundamentally crucial for cognitive functions related to learning and memory consolidation.
Participants reporting chronic pain in the absence of depressive indications demonstrated a discernible increase in hippocampal volume, alongside enhanced memory recall capabilities. This finding is congruent with the brain’s adaptive strategies to mitigate the physiological and psychological burden imposed by pain.
Conversely, individuals simultaneously afflicted by chronic pain and depression exhibited a reduction in hippocampal volume and a deficit in cognitive processing. Further dissection of these scans indicated that these structural and functional alterations manifested incrementally over time. This suggests an initial adaptive phase where the hippocampus endeavors to compensate for prolonged pain, followed by a gradual susceptibility to degradation as pain persists over extended durations.
Crucially, these observed neurobiological patterns were consistent across a diverse spectrum of chronic pain conditions, encompassing lumbar, abdominal, knee, and hip discomfort, as well as cephalalgia. This universality underscores that the findings are not confined to any singular type of chronic pain syndrome.
To further elucidate the sequence of these brain changes in individuals with chronic pain, we investigated analogous mechanisms using rodent models. This research revealed a parallel progression of alterations in hippocampal volume accompanied by heightened neuronal excitability in these animal subjects. Initially, moderate enhancements in cognitive function were noted, which subsequently yielded to anxiety-like behaviors, ultimately transitioning into depressive-like symptoms and diminished memory retention.
The hippocampus has long been recognized for its integral role in processing emotional memories and its pronounced vulnerability to the effects of chronic stress. The inherent plasticity of the hippocampus—its capacity for neurogenesis—is also understood to be a key component in managing prolonged stressful conditions.
Moreover, chronic stress has been implicated in accelerating neuronal apoptosis (programmed cell death) and suppressing adult neurogenesis—the continuous generation of new neurons within the hippocampus.
Our findings pinpointed a specific region within the hippocampus, known as the dentate gyrus—one of the few cerebral loci where new neurons are generated throughout adulthood—as the pivotal regulatory center orchestrating the transition from chronic pain to depression.
In the nascent stages of the pain experience, newly generated neurons in the dentate gyrus exhibited heightened activity, indicative of an initial protective response from the brain to sustained pain. However, with the passage of time, resident immune cells, termed microglia, became pathologically hyperactive, thereby disrupting the standard neural communication pathways within the hippocampus.
This aberrant activation of microglial cells appeared to signify a critical juncture, marking the point at which the brain’s initial compensatory mechanisms to pain began to falter.
Significantly, the administration of an antibiotic agent, minocycline, effectively attenuated the aberrant microglial activation and ameliorated depressive-like behaviors in our animal models. This therapeutic intervention also served to preserve hippocampal structural integrity and cognitive acuity.
Therapeutic Strategies for Pain and Depression
Our findings strongly suggest that therapeutic interventions like minocycline could be instrumental in averting the development of depression in individuals managing persistent pain, particularly if administered proactively.
It is important to acknowledge that a constellation of other influences, including psychosocial, socio-economic, and genetic factors, contribute to the subjective experience of pain perception. Consequently, it is plausible that these determinants may amplify chronic stress and pain perception in certain individuals.
Nevertheless, established evidence-based strategies exist to mitigate the risk of depression. A prior collaborative endeavor between Fudan University and the University of Cambridge demonstrated that adherence to seven healthy lifestyle parameters, encompassing adequate sleep, regular physical activity, and a balanced diet, could reduce the incidence of depression by an impressive 57%. Notably, these lifestyle factors were also correlated with augmented hippocampal volume, aligning seamlessly with our current research findings.
Mindfulness training presents another promising avenue. This approach emphasizes present-moment awareness and the minimization of distractions arising from intrusive thoughts and memories. The practice has been shown to enhance working memory capacity and promote increased hippocampal density.
A recent meta-analysis revealed that individuals proficient in mindfulness meditation exhibit enhanced grey matter volume, including within the hippocampus. Furthermore, the practice of mindfulness meditation has been demonstrated to correlate with increased hippocampal volume.
The beneficial effects of mindfulness practice extend to improving overall quality of life, not solely in the context of managing chronic pain, but also in alleviating symptoms of stress and depression.
Our recent discovery has successfully addressed a long-standing enigma that has perplexed researchers for years. We illuminated the pivotal role played by the brain’s hippocampus in elucidating why a segment of individuals experiencing chronic pain subsequently develop depression. This groundbreaking insight also illuminates potential therapeutic avenues for preventing depressive disorders in those afflicted by persistent pain.
The brain’s adaptive mechanisms we have identified may possess broader applicability to other conditions involving chronic stress, such as those arising from psychological trauma.![]()