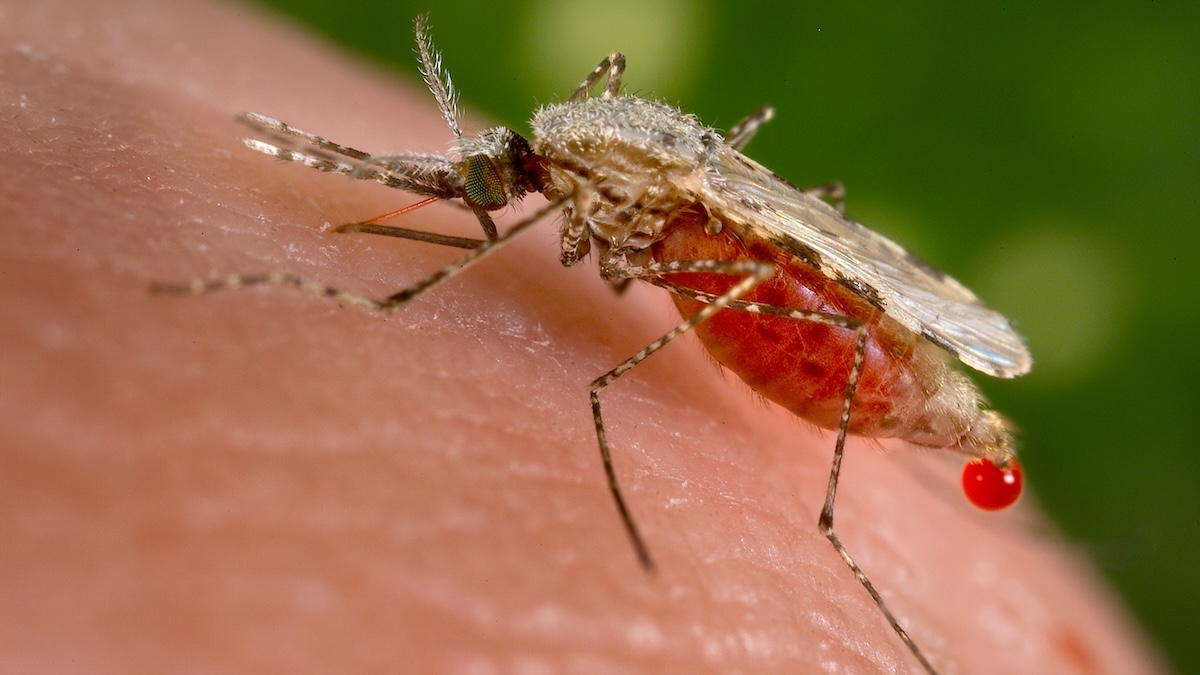
Approximately 610,000 individuals succumbed to malaria globally in 2024, with the African continent bearing the brunt of these fatalities, particularly among young children who remain the most susceptible demographic. In a significant development, scientific researchers have identified a potentially pivotal vulnerability within the pathogens responsible for this disease.
These groundbreaking findings offer profound insights into the intricate biological mechanisms of these parasites, as articulated by the creators of the recent study, and could pave the way for novel strategies to incapacitate them.
Precursors to current malaria-causing agents can be traced back to the Cretaceous period, and despite advancements in vaccine technology, this age-old affliction continues to plague human populations across increasingly expansive geographical areas.
Concurrently with ongoing vaccine development initiatives, the scientific community is actively seeking any exploitable weaknesses in either the parasites themselves or the mosquito vectors that facilitate their transmission to humans.
“The remarkable aspect of this discovery lies in the distinct nature of the malaria parasite’s ‘Aurora’ complex when contrasted with its counterpart found in human cells,” stated senior author Rita Tewari, a specialist in parasite cell biology at the University of Nottingham.
Malaria is instigated by protists, which are unicellular eukaryotic microorganisms that do not fall under the classifications of animals, fungi, or plants.
These particular protists belong to the genus Plasmodium, encompassing over 150 identified species that infect a variety of vertebrate hosts. While only five of these species are responsible for malaria in humans, their impact is profoundly destructive.
Plasmodium parasites exhibit rapid proliferation within human hosts and subsequently within Anopheles mosquitoes. A comprehensive understanding of the intricacies governing this accelerated process could prove immensely beneficial in our efforts to interrupt it.
The process of mitosis in malaria parasites is “fundamentally distinctive,” according to the study’s authors. These single-celled organisms undergo division and growth through a unique mechanism that deviates significantly from the typical processes observed in human cells and numerous other eukaryotic organisms.
The primary focus of the new investigation is on a protein known as Aurora-related kinase 1 (ARK1), which plays a crucial role in the unconventional cell division and developmental pathways of Plasmodium parasites.

Researchers have elucidated that ARK1 actively orchestrates events during the parasites’ mitosis, bearing the responsibility for assembling a specialized structure known as a spindle. This molecular apparatus is indispensable for the accurate segregation of genetic material, thereby enabling the generation of new parasites.
By employing advanced genetic manipulation techniques, specifically conditional gene knockout and gene knockdown, the research team successfully deactivated ARK1 within Plasmodium parasites to thoroughly assess its function.
The experimental outcomes strongly indicate that ARK1 functions as a critical vulnerability, akin to an Achilles’ heel, for malaria parasites. In the absence of this protein, the parasites were unable to form functional spindles, resulting in a failure of successful replication.
Parasites deficient in ARK1 were incapable of completing their developmental cycles within either vertebrate host cells or mosquitoes, thereby impeding their ability to propagate the disease.
In light of ARK1’s indispensable role for malaria parasites, this protein emerges as a highly promising candidate for the development of novel antimalarial therapeutic interventions, as indicated by the researchers.
“The designation ‘Aurora’ draws inspiration from the Roman goddess of dawn, and we are of the conviction that this protein truly signifies a new era in our comprehension of malaria cell biology,” remarked co-first author Ryuji Yanase, a cell biologist at the University of Nottingham.
Furthermore, owing to the fundamental disparities in our cellular machinery and biological processes compared to those of the parasites, targeting ARK1 could offer a strategic means of neutralizing malaria pathogens with minimal adverse effects on their human hosts.
“This distinction represents a significant advantage,” Tewari explained.
“It suggests the potential for developing pharmaceuticals specifically designed to target the parasite’s ARK1, effectively eradicating malaria without causing harm to the individual receiving treatment.”
By thoroughly documenting the unconventional methods of replication employed by these parasites, including the crucial involvement of ARK1, the authors aspire to establish a robust foundation for future scientific endeavors aimed at devising innovative strategies to counteract this persistent global health threat.