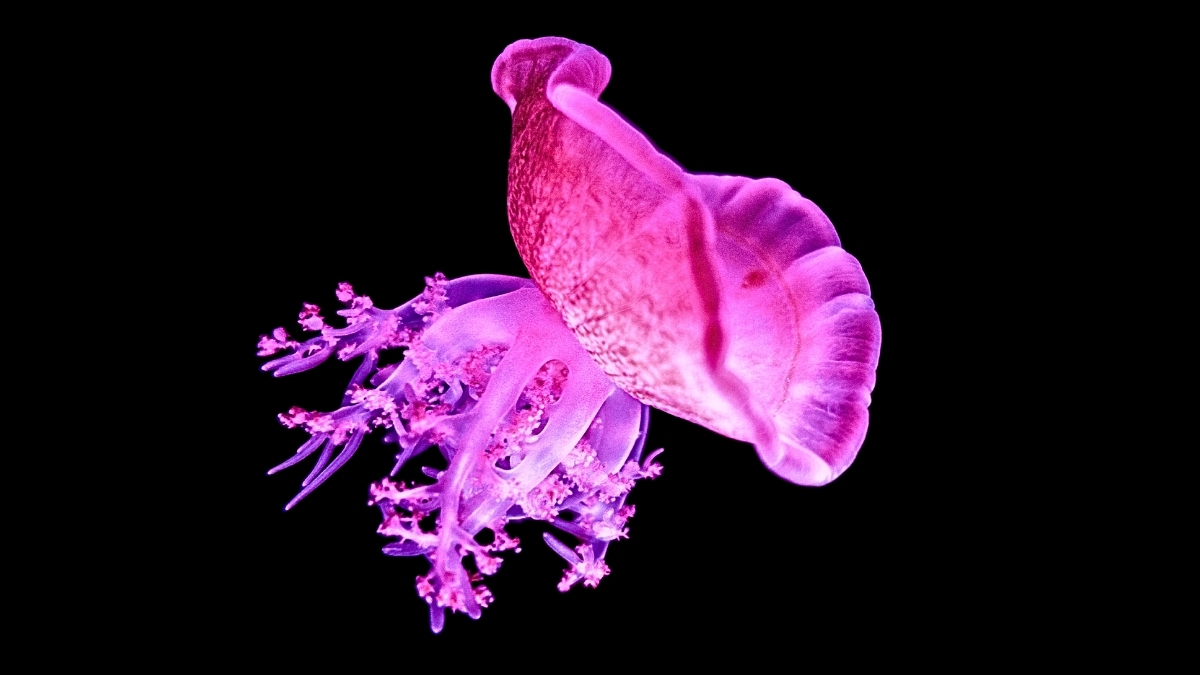
Even a rudimentary, gelatinous organism devoid of a specialized digestive exit and a central nervous system necessitates recuperative rest, according to novel research conducted by investigators at Bar-Ilan University in Israel.
Jellyfish dedicate approximately one-third of each diurnal cycle to slumber, mirroring human patterns, despite their markedly dissimilar biological makeup.
This finding implies that the genesis of sleep is profoundly ancient, considering that the lineage leading to humans diverged from the phylum encompassing jellyfish (Cnidaria) as far back as roughly a millennium ago.
Entities within the Cnidaria class lack a consolidated brain structure. Instead, they possess interconnected neural networks distributed along the entirety of their physical form. Notwithstanding this uncomplicated neurological architecture, these aquatic inhabitants have been observed to enter a state of torpor, much like other life forms equipped with nervous systems.
This period of quiescence and reduced responsiveness, however, is accompanied by inherent perils.
“The evolutionary emergence of sleep entailed significant trade-offs concerning fitness, such as diminished environmental awareness and increased susceptibility to predation,” articulate chronobiologist Raphaël Aguillon and his cohort in their published work.
Conversely, jellyfish typically slumber through the nocturnal hours akin to humans, and even engage in brief periods of rest during the midday. In contrast, their close relations, the sea anemones, adopt a nocturnal habit, sleeping throughout the daylight period. This suggests that sleep confers a potent advantage that effectively offsets its associated risks.
Specimens of the upside-down jellyfish (Cassiopea andromeda) and the starlet sea anemone (Nematostella vectensis) exhibited an escalation in neuronal DNA damage when subjected to sleep deprivation, a phenomenon the researchers documented under both controlled laboratory conditions and in their natural habitats.
Furthermore, when their surrounding conditions induced greater neuronal DNA damage, both Cnidaria species displayed an increased duration of sleep. These discoveries point towards sleep potentially having evolved as a protective mechanism against cellular deterioration.
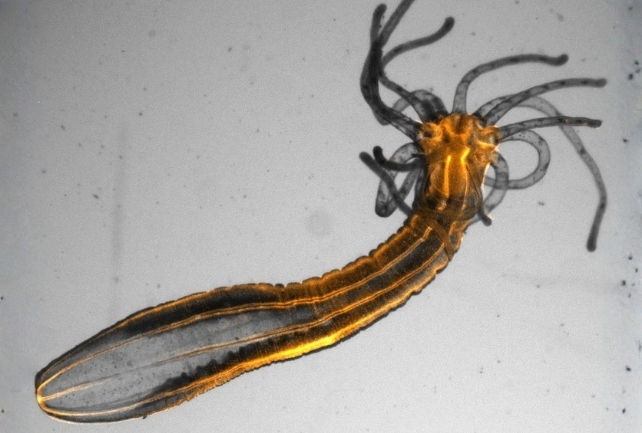
Upon administration of melatonin, the organisms experienced prolonged sleep periods, which subsequently led to a reduction in DNA damage. The researchers posit that Cnidarians utilize a melatonin system analogous to our own to regulate their sleep patterns in conjunction with diurnal cycles.
“Sleep deprivation, exposure to ultraviolet radiation, and the presence of mutagens all contributed to increased neuronal DNA damage and a heightened sleep drive,” the research team states in their publication.
“Both spontaneous and artificially induced sleep facilitated genomic stability.”
Consequently, even rudimentary nervous systems necessitate rest to mitigate the inherent cellular damage that accompanies periods of activity.
“The equilibrium between DNA damage and repair mechanisms is insufficient during states of wakefulness, and sleep provides a continuous interval that enables efficient cellular upkeep within individual neurons,” propose Aguillon, Harduf, and their collaborators.
“These findings indicate that the accumulation of DNA damage and cellular stress within primitive nerve nets may have been a driving force behind the evolutionary development of sleep.”