Ibuprofen, a widely recognized analgesic, is often the first recourse for alleviating discomfort ranging from common headaches to menstrual cramps.
However, recent scientific investigations suggest that this ubiquitous medication may offer benefits beyond merely assuaging pain, potentially possessing anticancer attributes.
As molecular scientists delve deeper into the intricate connections between inflammatory processes and the development of cancer, the role of ibuprofen is attracting considerable attention, prompting fascinating inquiries into how such a familiar pharmaceutical might confer unexpected protective effects.
Ibuprofen is classified within the cohort of non-steroidal anti-inflammatory drugs (NSAIDs). The association between NSAIDs and cancer prevention is not a novel concept; as early as 1983, clinical observations indicated that sulindac, an established prescription NSAIDakin to ibuprofen, was linked to a diminished incidence of colon cancer in specific patient cohorts.
Subsequent to these findings, researchers have actively explored the potential of these agents in thwarting or retarding the progression of other neoplastic diseases.
The mechanism of action for NSAIDs involves the inhibition of enzymes designated as cyclooxygenases (COX). Two primary isoforms exist. COX-1 is integral to maintaining the integrity of the gastric mucosa, preserving renal function, and facilitating hemostasis. Conversely, COX-2 is a key mediator in inflammatory cascades.
The majority of NSAIDs, including ibuprofen, exert inhibitory effects on both isoforms. This dual inhibition is the rationale behind the medical recommendation to administer these medications with food, thus mitigating potential gastrointestinal irritation.
Ibuprofen and Endometrial Malignancy
A comprehensive study published in 2025 demonstrated that ibuprofen may correlate with a reduced risk of endometrial cancer, the most prevalent form of uterine neoplasia, which originates in the uterine lining (endometrium) and predominantly affects postmenopausal women.
A significant modifiable risk factor for endometrial cancer is excess adiposity, as adipose tissue elevates circulating estrogen levels, a hormone known to promote the proliferation of malignant cells.
Additional risk determinants encompass advanced age, hormone replacement therapy (particularly estrogen-predominant regimens), diabetes mellitus, and polycystic ovary syndrome. Early menarche, delayed menopause, or nulliparity are also associated with increased susceptibility. Clinical manifestations may include aberrant vaginal hemorrhage, pelvic distress, and dyspareunia.
Within the framework of the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial, data pertaining to over 42,000 women aged 55–74 years were longitudinally analyzed over a 12-year period.
Participants reporting the consumption of at least 30 ibuprofen tablets per month exhibited a 25% reduction in the likelihood of developing endometrial cancer compared to those ingesting fewer than four tablets monthly. This observed protective effect was most pronounced among women with pre-existing cardiac disease.
Curiously, aspirin, another common NSAID, did not demonstrate a similar association with diminished risk in this particular investigation or in other published research. Conversely, aspirin has shown potential in preventing the recurrence of intestinal malignancies.
Other NSAIDs, such as naproxen, have been subjects of research concerning the prevention of colonic, vesical, and mammary cancers. The efficacy of these agents appears to be modulated by the specific cancer type, individual genetic makeup, and underlying physiological conditions.
Ibuprofen’s Broader Oncological Potential
The purported cancer-mitigating attributes of ibuprofen extend beyond endometrial neoplasia. Scientific literature indicates a potential reduction in the risk of developing colorectal, mammary, pulmonary, and prostatic cancers.
For instance, individuals with a history of previous colorectal cancer who utilized ibuprofen exhibited a lower probability of disease recurrence. Furthermore, its capacity to inhibit the proliferation and survival of colon cancer cells has been documented, and certain evidence even suggests a protective role against lung cancer in active smokers.
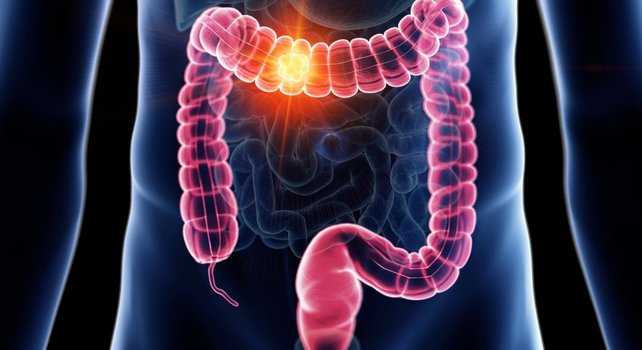
Inflammation serves as a cardinal characteristic of malignancy, and ibuprofen’s fundamental pharmacological action is its anti-inflammatory capacity.
By impeding COX-2 enzyme activity, the drug curtails the synthesis of prostaglandins, signaling molecules that propagate inflammation and cellular proliferation, including the aberrant growth of cancer cells. Reduced prostaglandin levels may therefore decelerate or arrest tumorigenesis.
However, this explanation represents only one facet of its multifaceted impact. Ibuprofen also appears to exert regulatory effects on cancer-associated genes such as HIF-1α, NFκB, and STAT3, which are instrumental in the survival of tumor cells under hypoxic conditions and contribute to therapeutic resistance.
Ibuprofen seems to diminish the transcriptional and functional activity of these genes, rendering malignant cells more susceptible. It can also modulate the supramolecular organization of DNA within cellular structures, potentially enhancing the sensitivity of cancerous cells to chemotherapeutic interventions.
A Note of Prudence
Nevertheless, the totality of scientific evidence is not uniformly conclusive. A study involving 7,751 patients indicated that the post-diagnosis administration of aspirin in cases of endometrial cancer was associated with increased mortality rates, particularly among individuals who had previously used aspirin. Other NSAIDs also appeared to elevate the risk of cancer-related demise.
Conversely, a recent systematic review concluded that NSAIDs, with a notable emphasis on aspirin, might reduce the incidence of several types of cancer. However, the sustained use of other NSAIDs could potentially escalate the risk of renal cell carcinoma. These disparate findings underscore the profound complexity of the interplay between inflammatory pathways, immune responses, and oncogenesis.
Despite the promising implications, medical professionals strongly advise against self-prescribing ibuprofen for the express purpose of cancer prophylaxis. Prolonged or high-dose utilization of NSAIDs can precipitate severe adverse events, including but not limited to gastric ulcerations, gastrointestinal hemorrhage, and renal impairment.
Less frequently, these agents may precipitate cardiovascular incidents such as myocardial infarctions or strokes. Furthermore, NSAIDs exhibit potential for drug interactions with numerous pharmaceuticals, including warfarin and certain classes of antidepressants, thereby augmenting the risk of hemorrhagic complications and other untoward effects.
The prospect that a common analgesic could contribute to cancer prevention is both exhilarating and thought-provoking. Should subsequent research corroborate these preliminary findings, ibuprofen might eventually be integrated into a comprehensive strategy for mitigating cancer risk, particularly within high-risk demographics.
Presently, the consensus among experts favors prioritizing lifestyle-driven preventive measures: adherence to an anti-inflammatory dietary pattern, maintenance of a healthy body mass index, and consistent engagement in physical activity.
While everyday medications may indeed harbor unforeseen therapeutic promise, until definitive scientific validation is established, the most time-honored prescription for cancer risk reduction remains paramount: embrace a balanced diet, cultivate an active lifestyle, and always seek professional medical guidance before resorting to pharmacological interventions.
Dipa Kamdar, Senior Lecturer in Pharmacy Practice, Kingston University; Ahmed Elbediwy, Senior Lecturer in Cancer Biology & Clinical Biochemistry, Kingston University, and Nadine Wehida, Senior Lecturer in Genetics and Molecular Biology, Kingston University