The noxious secretion of black widow arachnids comprises a sophisticated array of seven distinct latrotoxins. However, only one, specifically α-latrotoxin, exhibits predatory efficacy against vertebrates, encompassing humankind. Researchers specializing in chemistry at the University of Münster have successfully elucidated the structural configuration of α-latrotoxin, both in its pre- and post-membrane insertion states, at an almost atomic level of detail.
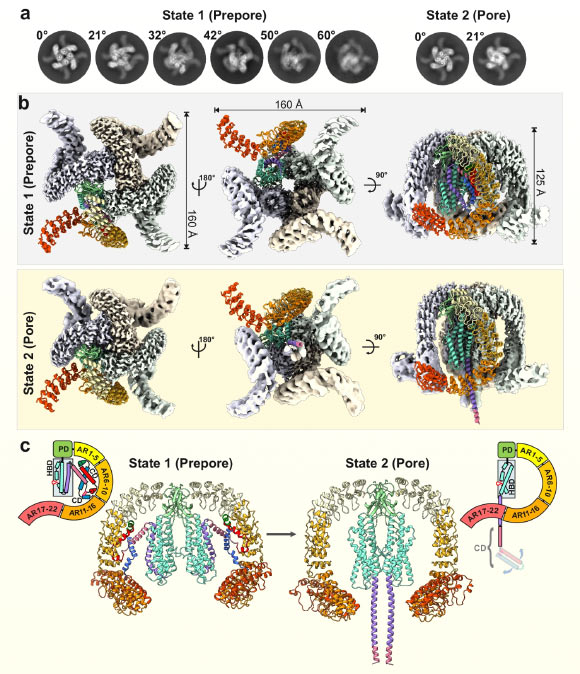
Cryo-EM structure of α-latrotoxin in two distinct tetrameric states. Image credit: Klink et al., doi: 10.1038/s41467-024-52635-5.
Latrotoxins constitute the primary detrimental constituents within the venom produced by black widow spiders, belonging to the genus Latrodectus.
This complex venom includes the α-latrotoxin, which is exclusively pathogenic to vertebrates, alongside five insecticidal agents designated as α, β, γ, δ, and ε-latroinsectotoxins, in addition to a solitary toxin specialized for crustaceans.
“α-Latrotoxin disrupts the intricate process of signal transmission within the nervous system,” stated Björn Klink, a researcher at the University of Münster, in conjunction with his colleagues.
“Upon initial engagement of α-latrotoxin with specific receptors situated on the synapses—the critical junctions between neurons or between neurons and muscular tissues—a drastic influx of calcium ions occurs, overwhelming the presynaptic membranes of the cells responsible for signaling.”
“This aberrant calcium influx precipitates the expulsion of neurotransmitters, thereby initiating severe muscular contractions and convulsive spasms.”
“Notwithstanding the apparent straightforwardness of this biological event, an extraordinarily sophisticated molecular mechanism underlies its execution.”
To achieve a more profound comprehension of the process governing the cellular ingress of calcium into the presynaptic membrane, the research team employed advanced cryo-electron microscopy (cryo-EM) techniques and sophisticated molecular dynamics (MD) computational simulations.
Their investigations revealed that α-latrotoxin undergoes a profound structural metamorphosis upon interaction with its target receptor.
A segment of the toxic molecule reconfigures to form a columnar structure, which then penetrates the cellular membrane in a manner analogous to a hypodermic needle.
A notable characteristic of this structure is its capability to generate a minute conduit within the membrane, which subsequently functions as a channel facilitating the passage of calcium ions.
The MD simulations provided critical insights, demonstrating that calcium ions are able to traverse into the cell via a highly selective aperture located on the side of the conduit, positioned directly above the main pore.
“The toxin ingeniously emulates the functional role of the native calcium channels present in the presynaptic membrane through an exceptionally elaborate mechanism,” commented Christos Gatsogiannis, another researcher from the University of Münster.
“Consequently, it presents a unique profile, distinguishing itself in every discernible aspect from any toxins previously documented.”
“These novel discoveries unlock a broad spectrum of promising potential applications.”
“Latrotoxins possess substantial biotechnological promise, paving the way for advancements such as the creation of more effective antidotes, innovative therapeutic interventions for paralytic conditions, and the development of novel bio-based pesticides.”
The comprehensive research has been formally published in the esteemed scientific journal, Nature Communications.
_____
B.U. Klink et al. 2024. Structural basis of α-latrotoxin transition to a cation-selective pore. Nat Commun 15, 8551; doi: 10.1038/s41467-024-52635-5