New scientific findings suggest that certain genetic variations, present in approximately 10% of the populace, may diminish the efficacy of GLP-1 treatments for type 2 diabetes, such as Ozempic and Wegovy.
These therapeutic agents are named in homage to the glucagon-like peptide-1 (GLP-1) hormone they mimic. For individuals managing diabetes, this involves regulating glucose levels by enhancing insulin secretion and decelerating the digestive process.
However, the therapeutic impact of GLP-1 medications on type 2 diabetes patients exhibits considerable variability. An international consortium of researchers embarked on a new investigation to ascertain whether genetic factors contribute to this disparity.
To probe this phenomenon, the research team concentrated on an enzyme known as PAM (an acronym for peptidyl-glycine alpha-amidating monooxygenase) and the gene responsible for its synthesis.
Roughly one in ten individuals carry variations within the PAM gene that can impede hormone activation, including that of GLP-1. These PAM variants are also observed with greater frequency among individuals diagnosed with diabetes.
“In my clinical practice with diabetes patients, I observe a substantial variability in how individuals respond to these GLP-1 based medications, and this response is challenging to predict clinically,” stated endocrinologist Mahesh Umapathysivam, affiliated with Adelaide University in Australia.
“This research marks a foundational step towards leveraging an individual’s genetic profile to refine therapeutic decision-making.”
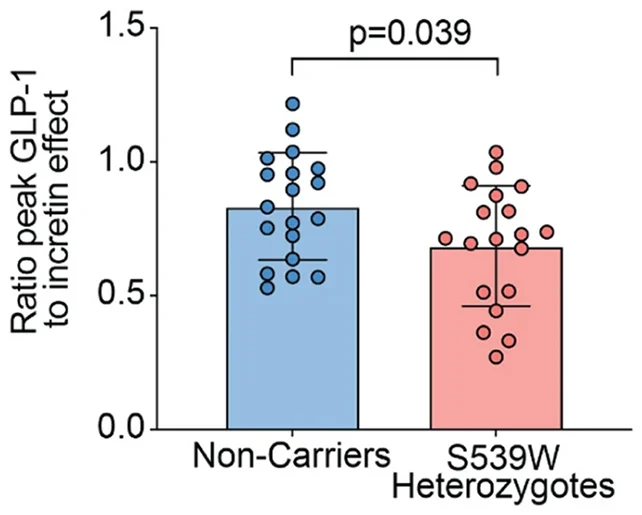
In their analytical endeavor, the scientists conducted a comparative study involving 19 individuals with the PAM gene variant p.S539W against 19 carefully matched control subjects, observing their physiological reactions after consuming a sugar-rich beverage.
Intriguingly, the cohort with the genetic variant exhibited elevated GLP-1 levels. It was discovered that the p.S539W variation induced GLP-1 resistance, thereby hindering the hormone’s appropriate metabolic processing.
“Despite the presence of higher circulating GLP-1 concentrations in individuals with the PAM variant, we found no indication of enhanced biological activity,” commented endocrinologist Anna Gloyn from Stanford University.
“Their blood sugar levels did not decrease at an accelerated rate. A greater quantity of GLP-1 was necessitated to elicit an equivalent biological effect, signifying resistance to GLP-1.”
This unexpected metabolic shift prompted further investigations, including experiments on mouse models where the PAM gene was rendered inactive. This genetic manipulation resulted in the absence of PAM enzyme activity, leading to the development of GLP-1 resistance in the mice, with their blood glucose levels remaining unregulated despite elevated GLP-1 concentrations.
The investigational scope was further broadened. The subsequent phase involved a comprehensive analysis of clinical trials encompassing a total of 1,119 participants, aimed at determining if individuals with specific PAM variations displayed a less favorable response to GLP-1 medications.
Once again, the findings corroborated the hypothesis that these genetic variations correlate with GLP-1 resistance: individuals possessing the standard PAM gene variants exhibited superior responses to GLP-1 therapy compared to those who did not. This specific genetic association was not observed in relation to other evaluated diabetes medications.
These findings open significant avenues for future scientific inquiry. The precise biological mechanisms underlying the blunted effect of GLP-1 drugs remain a subject requiring detailed elucidation.
Researchers may also explore the interplay between PAM genes and GLP-1 medications utilized for weight management, which typically involve higher dosages.
Prospectively, genetic screening could potentially ascertain the likelihood of GLP-1 treatments being effective for individuals with type 2 diabetes, thereby reducing the expenditure of time and financial resources on suboptimal pharmacological interventions.
Furthermore, there is potential for the modification of GLP-1 drugs to overcome the impact of these PAM gene variations, although this remains an area in its nascent stages of development.
“It is a common practice for pharmaceutical entities to gather genetic data from their study participants,” noted Gloyn.
“Given the existence of a broad category of medications known as insulin sensitizers, it may be feasible to develop therapeutic agents that enhance individuals’ sensitivity to GLP-1s or to devise GLP-1 formulations, such as extended-release versions, that circumvent GLP-1 resistance.”