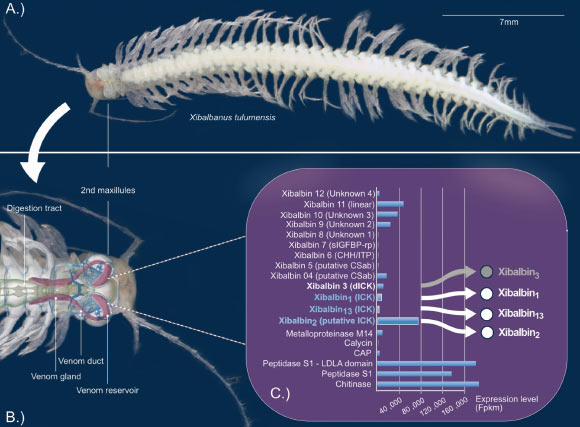
The sole crustacean with a documented venom system is Xibalbanus tulumensis, a venomous remipede inhabiting anchialine caverns on Mexico’s Yucatán Peninsula.
“Venomous organisms deploy their toxic compounds into other organisms predominantly for self-preservation or to capture prey,” stated Dr. Björn von Reumont, a researcher affiliated with Goethe University Frankfurt, alongside his colleagues.
“A multitude of venoms consist of proteins that have undergone evolutionary development to influence a spectrum of physiological functions within their target organisms.”
“The exploration of these bioactivities holds the potential for applications in pharmacology or agrochemistry.”
“The vast majority of investigated venoms and venom proteins predominantly originate from prominent terrestrial groups, including snakes, spiders, scorpions, and insects,” they elaborated.
“Marine fauna has attracted comparatively less research scrutiny, with only a limited number of fish and invertebrate species—such as sea anemones, jellyfish, cone snails, cephalopods, polychaetes, and more recently, nemerteans—having been subjected to more thorough study.”
“Given that venoms and their constituent toxic proteins have evolved independently across diverse animal lineages, the investigation of novel lineages offers, on one hand, an avenue for identifying previously unknown venomous compounds possessing intriguing bioactivity, and on the other, an opportunity to deepen our comprehension of the evolutionary mechanisms behind convergent functional traits in general.”
In their recent research endeavor, scientists meticulously examined the bioactivity of peptides identified within the venom of the crustacean species Xibalbanus tulumensis.
This subterranean, cave-dwelling crustacean belongs to the class Remipedia, a group initially described in the 1980s and currently encompassing 28 extant species.
“Xibalbanus tulumensis inhabits the cenotes, which are submerged cave systems found on Mexico’s Yucatán Peninsula,” the researchers noted.
“This troglobitic crustacean directly injects the venom synthesized in its venom gland into its prey.”
“This potent toxin is composed of a variety of elements, including a novel peptide class, which has been christened xibalbine, in honor of its crustacean source.”
“Certain xibalbines feature a distinctive structural motif observable in other venoms, particularly those produced by arachnids: several amino acid residues (cysteines) within the peptide are interconnected in a manner that forms a complex, knot-like configuration.”
“This structural arrangement confers remarkable resilience upon the peptides, rendering them resistant to enzymatic degradation, thermal denaturation, and extreme pH conditions.”
“Such ‘knottins’ frequently function as neurotoxins, exerting their effects by interacting with ion channels and inducing paralysis in prey—a mode of action also hypothesized for some xibalbines.”
The experimental findings demonstrate that all xibalbine peptides subjected to evaluation by the research team—with specific emphasis on Xib1, Xib2, and Xib13—exhibited pronounced inhibitory effects on potassium channels within mammalian physiological systems.
“This inhibitory capacity holds significant implications for the development of therapeutic agents targeting a spectrum of neurological disorders, including epilepsy,” remarked Dr. von Reumont.
“Furthermore, Xib1 and Xib13 displayed the capability to impede the function of voltage-gated sodium channels, akin to those present in neural and cardiac muscle tissues.”
“Beyond this, within the sensory neurons of higher mammals, these two peptides were observed to activate two crucial signaling proteins—kinases PKA-II and ERK1/2—which play integral roles in cellular signal transduction pathways.”
“The latter observation suggests a potential involvement in pain sensitization mechanisms, thereby illuminating novel therapeutic avenues for pain management.”
The comprehensive research findings generated by the scientific team were formally disseminated in the esteemed journal BMC Biology.
_____
E.L. Pinheiro-Junior et al. 2024. Diversely evolved xibalbin variants from remipede venom inhibit potassium channels and activate PKA-II and Erk1/2 signaling. BMC Biol 22, 164; doi: 10.1186/s12915-024-01955-5