While Alzheimer’s disease is commonly perceived as an ailment predominantly affecting older individuals, the pathological transformations within the brain that define it actually commence much earlier, approximately around the third decade of life.
The initial stages of these cerebral alterations involve the aberrant aggregation of a protein known as tau. This abnormal tau begins accumulating in a diminutive, deep-seated brain region responsible for regulating sleep, attention, and wakefulness – the locus coeruleus. Subsequently, this abnormal tau disseminates throughout the wider brain.
The formation of tau tangles does not inherently indicate the presence of Alzheimer’s disease; indeed, this phenomenon occurs to varying extents in nearly all individuals. However, given that these pathological changes originate in the locus coeruleus, some neuroscientists, including myself, regard this area as a critical early indicator, akin to a harbinger for the development of Alzheimer’s disease.
Our research is focused on investigating whether impeding or decelerating the formation of tau tangles within this specific brain region, or preserving its overall health through other means, could serve as a viable strategy to disrupt the progression of the disease and mitigate age-related cognitive decline.
Current investigations emanating from my laboratory and other research institutions are exploring the potential of a therapeutic intervention known as vagus nerve stimulation. This modality, already widely employed for various health conditions, is being examined as a potential means to ensure the optimal functioning of the locus coeruleus.
The locus coeruleus and Alzheimer’s disease
Situated within the brainstem, the most caudal portion of the brain, lies the locus coeruleus. Its nomenclature, translating to “blue spot,” derives from the presence of a pigment called neuromelanin, which is synthesized by its constituent cells.
The locus coeruleus assumes a pivotal role in numerous facets of fundamental human physiology. It is accountable for producing nearly all of the brain’s norepinephrine, a crucial neurotransmitter indispensable for regulating sleep quality, alertness levels, attentional capacity, concentration, learning processes, and even immune system responsiveness.
Furthermore, it receives afferent signals from neural pathways originating throughout the brain and peripheral nervous system— including those from the vagus nerve, which transmits bidirectional information to and from the cardiovascular, respiratory, and other organ systems.
My research endeavors to elucidate the intricacies of this brain region’s architecture, the mechanisms by which neuronal signal transmission occurs within its confines, and its interconnections with other neural networks. I also examine how these structural and functional attributes evolve across the lifespan and their impact on cognitive functions such as thought and memory.
Evidence from numerous studies suggests that commencing in middle age, neurons within the locus coeruleus may become compromised by the accumulation of tau protein, and this neuronal damage may exhibit a correlation with diminished memory performance. The pathological cascade of tau aggregation, neuronal demise, and functional impairment within the locus coeruleus demonstrably precedes and serves as a predictive marker for Alzheimer’s diagnosis and the manifestation of its symptoms.
This realization has prompted researchers to posit that maintaining the integrity and health of the locus coeruleus could offer protective benefits for the remainder of the brain as well.
Vagus nerve stimulation and brain health
During the 1980s and 1990s, pioneering research revealed that the application of vagus nerve stimulation could effectively alleviate seizure frequency in epilepsy. Concurrently, it was observed that this intervention frequently conferred additional advantageous effects, such as enhancements in mood regulation and cognitive function.
Currently, vagus nerve stimulation has received regulatory approval from the Food and Drug Administration for applications beyond epilepsy treatment, encompassing its use in managing migraine headaches, depressive disorders, and facilitating recovery post-stroke.
The administration of vagus nerve stimulation for conditions like epilepsy and depression typically involves the surgical implantation of an electrical stimulator device in the left pectoral region, where the vagus nerve is accessible. Non-invasive methods for headache treatment involve delivering gentle electrical impulses to specific points on the neck or ear, areas where the vagus nerve courses in close proximity to the skin’s surface.
Prior to the establishment of the locus coeruleus’s definitive link to Alzheimer’s disease, researchers had already theorized that vagus nerve stimulation might exert beneficial effects on mood and cognition in individuals afflicted with the condition. This hypothesis was predicated on the understanding that vagus nerve stimulation could, in part, potentiate norepinephrine levels in the brain—a neurotransmitter that is demonstrably deficient in individuals with Alzheimer’s disease within their cerebral environments.
Maintaining Optimal Function
Neuroscientists are still endeavoring to fully unravel the precise mechanisms by which vagus nerve stimulation confers its neuroprotective benefits. However, a prominent theoretical framework suggests that it plays a role in modulating the intrinsic activity patterns of neurons within the locus coeruleus, thereby promoting its proper physiological operation.
An excessively activated locus coeruleus could potentially induce hyperarousal, leading to feelings of intense stress or even panic. In effect, a hyperactive locus coeruleus is implicated in the exacerbation of certain symptomatology associated with post-traumatic stress disorder. Conversely, a hypoactive locus coeruleus can contribute to depressive states or memory impairments.
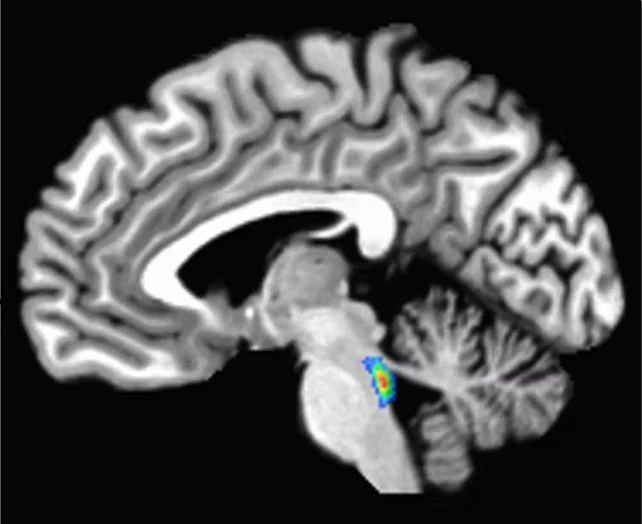
Certain modalities of vagus nerve stimulation appear to exert their effects not by directly increasing or decreasing locus coeruleus activity, but rather by influencing the temporal dynamics and firing rhythm of its neuronal population. Other forms of vagus nerve stimulation have been observed to elevate norepinephrine levels in the brains of rodent models, leading researchers to hypothesize that this mechanism might also underpin its therapeutic efficacy in treating epilepsy.
These varied experimental findings have led to the proposition that vagus nerve stimulation might function as a sophisticated regulatory agent for the locus coeruleus, enabling it to achieve and sustain optimal activity levels for peak physiological performance.
Can Vagus Nerve Stimulation Mitigate Memory Deficits?
Encouraging preliminary evidence suggests that vagus nerve stimulation may offer protective benefits for the aging brain.
A growing body of research indicates that vagus nerve stimulation can prevent the deterioration of memory function or even facilitate its improvement in individuals diagnosed with mild cognitive impairment or those in the nascent stages of Alzheimer’s disease.
One notable clinical trial involving 52 participants, aged between 55 and 75, who had received a diagnosis of mild cognitive impairment, reported statistically significant enhancements in memory recall and overall cognitive performance following a six-month regimen of vagus nerve stimulation administered for one hour daily, five days per week during the intervention period.
Furthermore, research conducted on healthy older adults in their sixties, as well as on healthy young adults aged 18 to 25, has reported improvements in diverse aspects of memory function following even a single session of vagus nerve stimulation.
While this line of inquiry remains in its nascent stages, it offers a promising avenue for developing novel strategies to mitigate some of the debilitating symptoms associated with Alzheimer’s disease and the natural processes of cognitive aging.![]()