The emergence of messenger RNA (mRNA) vaccines against SARS-CoV-2 in 2020 fundamentally altered the trajectory of the COVID-19 pandemic. Presently, this Nobel Prize-winning technological innovation is being repurposed for oncological applications, with mRNA vaccines undergoing clinical evaluation for conditions such as melanoma, small cell lung cancer, and bladder cancer. This development portends novel strategies for both the prevention and therapeutic management of malignancy.
Initial scientific conjecture proposed that a singular, specific immune cell subtype was indispensable for mRNA vaccination to elicit an immune system response. However, a recent investigation conducted by researchers at Washington University School of Medicine in St. Louis, utilizing murine models, demonstrates that even in the absence of these particular cells, mRNA vaccines can still instigate robust anti-tumor immunogenic activity. The underlying mechanism, as uncovered by the study, involves a related immune cell subtype that can also stimulate anti-tumor immunity – a finding that carries particular significance given this analogous cell type’s lack of involvement in responses to other vaccine modalities.
The ramifications of these findings, disseminated on April 15th in the esteemed journal Nature, offer a more profound comprehension of the immune system’s interaction with mRNA vaccination and provide crucial guidance for the optimal architecture of cancer vaccines.
There is substantial enthusiasm surrounding the application of mRNA vaccine methodologies, successfully employed during the COVID-19 pandemic, to the challenge of inducing anti-tumor immunity. By meticulously examining the implicated immune cells and the intricate coordination of their responses, we are furnishing vaccine developers with supplementary mechanistic insights to consider as they strive to optimize these vaccines against tumor-specific antigens.”
Kenneth M. Murphy, MD, PhD, senior author, the Eugene Opie Centennial Professor of Pathology & Immunology at WashU Medicine
Dr. Murphy also holds a research affiliation with the Siteman Cancer Center, which operates under the auspices of Barnes-Jewish Hospital and WashU Medicine.
An Unconventional Immunological Pathway
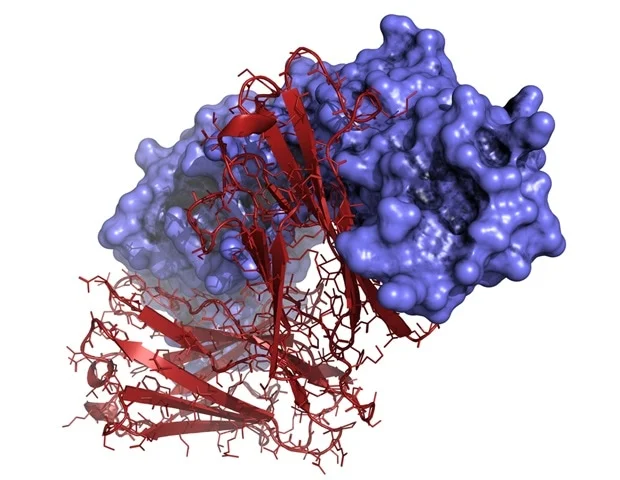
mRNA vaccines function by conveying genetic blueprints, encapsulated within messenger RNA biomolecules, that instruct immune cells to synthesize protein fragments. These fragments, in turn, stimulate the immune system to identify and eliminate cells that exhibit these proteins. Dendritic cells, a specialized immune cell population, are responsible for translating the mRNA directives into protein components. Subsequently, T cells, another crucial immune cell type, undertake the task of locating and neutralizing these targeted cells. By designing mRNA vaccines to express protein fragments unique to a tumor, T cells can be effectively directed to eradicate cancerous growths.
Historically, classical type 1 dendritic cells (cDC1) have been recognized for their proficiency in educating T cells to combat viral infections. However, the precise mechanisms by which T cells achieve activation following mRNA vaccination, whether against viral pathogens or neoplastic cells, remain less clearly elucidated. In a collaborative effort with Dr. William E. Gillanders, the study’s co-corresponding author and the Mary Culver Professor of Surgery at WashU Medicine, Dr. Murphy and his laboratory team employed murine models engineered to lack either cDC1 or a closely related dendritic cell subtype, known as cDC2. This experimental design allowed for the meticulous dissection of the roles played by different dendritic cell populations in the priming of T cells subsequent to mRNA cancer vaccination.
Dr. Gillanders, a physician-scientist and surgical oncologist who has also been instrumental in the development of an investigational vaccine for triple-negative breast cancer, provides clinical care to patients at the Siteman Cancer Center.
As an integral component of this research initiative, the investigators observed that mice inoculated with an mRNA vaccine exhibited robust T-cell responses, even when cDC1 cells were absent. Furthermore, these immunized mice, lacking cDC1s, demonstrated an ability to clear sarcoma tumors – a category of cancers originating in connective tissues such as adipose, muscular, neural, vascular, osseous, and cartilaginous structures. This observation strongly implied that an alternative cellular entity must be facilitating the T-cell activation cascade.
Indeed, the study confirmed that cDC2 cells also actively contribute to the genesis of T-cell-mediated immune responses and play a role in inhibiting tumor progression. An additional significant finding of the study was that T cells activated by cDC1s and cDC2s displayed subtly distinct molecular “fingerprints.” These differentiating characteristics could prove invaluable for scientists engaged in the design of more efficacious future vaccine iterations.
Concurrently, immunized mice deficient in cDC2s, as well as those possessing both dendritic cell subtypes, generated comparable immune responses and exhibited tumor rejection. This collective outcome underscores the fact that mRNA vaccination leverages the synergistic action of both dendritic cell subtypes to impede malignant growth.
In-depth investigation into the function of cDC2s revealed that they stimulate T cells through an indirect mechanism, often referred to as “cross-dressing.” This process involves the outsourcing of tasks to other cells, which interpret the mRNA instructions to produce the relevant protein. These intermediate cells then fragment the protein and present minute peptides on their surface. Subsequently, these cells transfer the cell membrane complex bearing the presented peptide to the cDC2, enabling the cDC2 to interact with T cells – a phenomenon previously documented as “cross-dressing.”
“This research illuminates a novel mechanism by which mRNA vaccines engage the immune system – through the concerted action of both cDC1 and cDC2. This discovery serves to elucidate their potency and provides researchers with concrete targets for enhancing the efficacy of future mRNA cancer vaccines,” stated Dr. Gillanders. “This could lead to refinements in vaccine formulation and dosage, potentially shed light on inter-individual variations in vaccine responsiveness, and guide strategies aimed at maximizing vaccine effectiveness.”
Jo, S. et al. (2026). mRNA vaccines engage unconventional pathways in CD8+ T cell priming. Nature. DOI: 10.1038/s41586-026-10353-6. https://www.nature.com/articles/s41586-026-10353-6