A recent scientific investigation has illuminated a significant transformation in female mammary tissue as it advances in age, fostering conditions conducive to the proliferation of neoplastic cells.
A collaborative effort between researchers affiliated with the University of Cambridge in the United Kingdom and the University of British Columbia in Canada has culminated in the creation of the most comprehensive atlas of mammary tissue alterations documented to date.
This meticulously detailed map, encompassing an expanse of over three million individual cells, chronicles the evolutionary trajectory of breast tissue throughout a woman’s lifespan, pinpointing a particularly pronounced shift around the menopausal transition.
“Our analysis revealed that as women mature, their mammary tissue undergoes substantial modifications, with the most profound changes manifesting during menopause,” stated Pulkit Gupta, a specialist in oncological research and a co-lead author of the study, who is presently affiliated with Vanderbilt University.
“While alterations are also observed in a woman’s twenties, potentially in connection with gestation and parturition, these are considerably less pronounced.”
The researchers ascertained that as women enter later stages of life, there is a reduction in the cellular density within breast tissue, accompanied by structural modifications. These environmental shifts are believed to facilitate the survival and propagation of malignant cells, thereby elucidating the age-related escalation of breast cancer susceptibility.
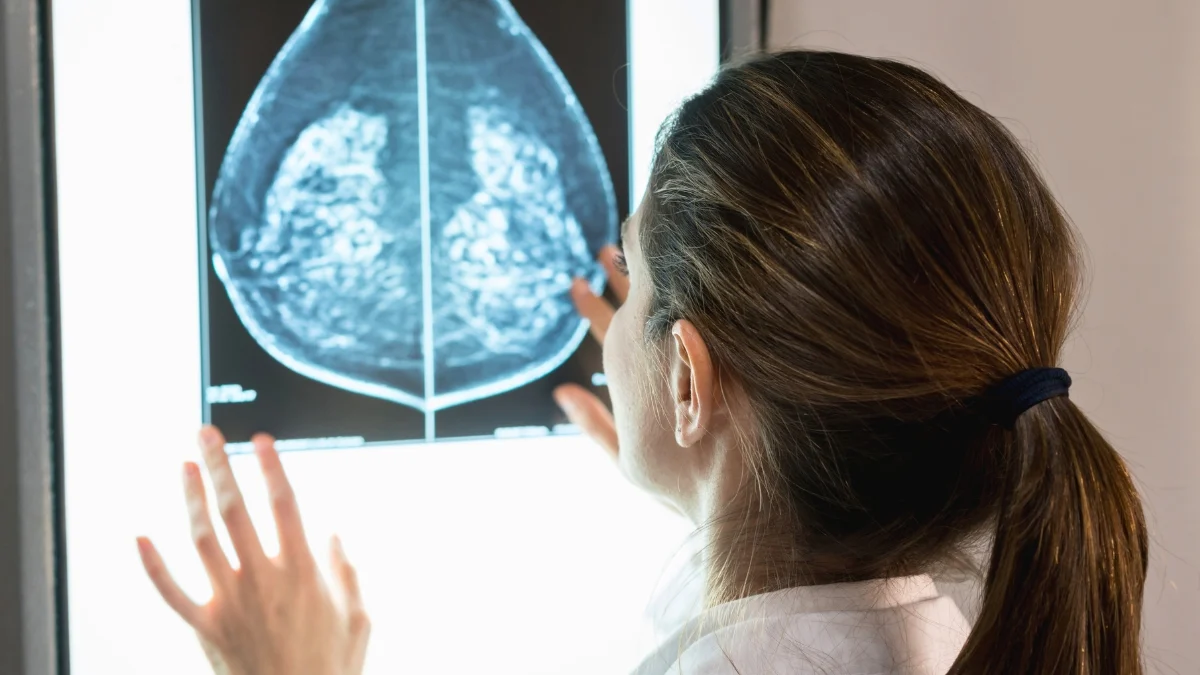
An estimated 320,000 women in the United States are projected to receive a breast cancer diagnosis this year, according to the American Cancer Society, positioning it as the most prevalent malignancy among females.
An upward trend in breast cancer incidence is being observed among younger women, the underlying reasons for which remain somewhat elusive, though the majority of diagnoses still occur in women aged 50 and above.
Advanced age stands as one of the primary determinants of risk for a vast spectrum of oncological conditions, including breast cancer. This phenomenon is partly attributable to the cumulative accumulation of genetic anomalies within cells over time, leading to aberrant cell development that can proliferate uncontrollably and manifest as neoplasms.
However, the intricate mechanisms by which mammary tissue ages and how these alterations contribute to oncogenesis remain subjects of considerable scientific inquiry.
“Despite the fact that breast cancer affects well over two million women globally, our understanding of its etiology and temporal occurrence is notably limited,” commented Gupta.
To address this knowledge gap, the investigators meticulously examined samples of healthy breast tissue obtained from 527 women, aged between 15 and 86 years, who underwent elective breast reduction surgery.
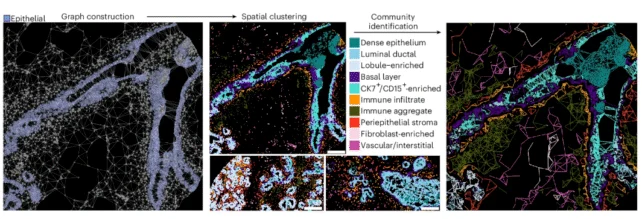
Employing sophisticated imaging methodologies, the research team meticulously charted individual cellular components, thereby generating a high-resolution two-dimensional representation of mammary tissue evolution through the passage of time.
The resultant map indicated a diminished rate of cellular proliferation with advancing age, alongside a reduction in the populations of immunocytes and stromal cells (responsible for providing structural support to the tissue). Concurrently, there was a decrease in the number of epithelial cells, which constitute the lining of the mammary ducts and the lobules responsible for milk production.
“It is not unexpected to observe a decline in epithelial cell count, given their role in lactation, a function that assumes less prominence with age,” observed Raza Ali, a consultant pathologist and co-senior author from the University of Cambridge.
“Nevertheless, the sheer magnitude of these widespread mammary tissue changes was indeed surprising.”
The architecture of the breast tissue itself underwent a metamorphosis; the milk-producing lobules exhibited involution or complete regression, an augmentation in adipocytes was noted, and a diminution in vascularization occurred.
Furthermore, a spatial redistribution of immune and stromal cells was observed, with these cells migrating further from epithelial cells, potentially facilitating the escape and dissemination of neoplastic elements.
In younger mammary glands, there was a greater abundance of specific immune cells, namely B-cells and activated T-cells, which play a crucial role in the immunosurveillance and elimination of nascent malignant cells.
Conversely, older mammary tissues were characterized by a deficit in these cancer-fighting cellular populations and an enrichment in M2 macrophages, a type of immune cell that prior research has implicated in the pathogenesis of cancer.
These transformations collectively foster a more pro-inflammatory milieu, wherein the immune system’s capacity to contain oncological progression is likely compromised, thereby becoming “more permissive of carcinogenesis,” as the researchers articulated in their publication.
“The precise determinants for these shifts in immune cell populations remain unelucidated,” opined Ali.
“We can hypothesize that one contributing factor may be the high concentration of immunoglobulins present in breast milk, likely intended to bolster infant immunity, and these are elaborated by B cells.”
“What is unequivocally evident from our detailed mapping,” Ali added, “is that all these observed modifications collectively establish an environment where spontaneously arising cancer cells encounter diminished resistance with advancing age, facilitating their establishment and dissemination.”
The investigators identified a singular, substantial phase of aging occurring in the late forties, coinciding with the menopausal period.
The rate at which mammary tissue undergoes aging is likely subject to individual variability, and such discrepancies are anticipated to influence an individual’s predisposition to cancer, the authors observed.
The scope of this particular study did not extend to an analysis of ethnicity, genetic predispositions, or other established risk factors for breast cancer. However, prior investigations have indicated that the normal breast tissue of women carrying genetic predispositions for breast cancer exhibits indicators of premature aging.
This groundbreaking research has been disseminated in the esteemed journal Nature Aging.