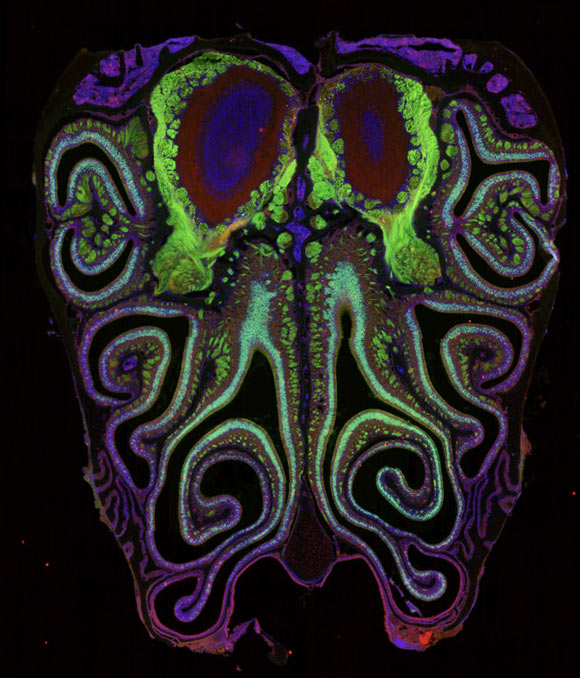
By meticulously charting millions of olfactory sensory neurons within rodent models, researchers have unveiled intricate, striped formations within the nasal cavity, fundamentally challenging long-standing hypotheses regarding olfactory neural architecture.
“For the majority of individuals, the faculty of smell represents an indispensable facet of daily experience,” articulated Professor Sandeep (Robert) Datta from Harvard Medical School.
“It furnishes vital intelligence concerning our surrounding environment, serves as a crucial alert mechanism for potential hazards, amplifies gustatory perception, and possesses the remarkable capacity to trigger emotional responses and recollections.”
“Nevertheless, from a scientific vantage point, the mechanisms of olfaction remain profoundly enigmatic.”
In their investigation utilizing mice, Professor Datta and his research cohort ascertained that contrary to prevailing scientific understanding, the neurons responsible for expressing these olfactory receptors exhibit a pronounced degree of spatial organization: they are arrayed in horizontal bands corresponding to specific receptor types, spanning from the uppermost to the lowermost regions of the nasal passage.
Furthermore, their findings demonstrate a congruency between the topographical arrangement of receptors within the nose and the corresponding olfactory maps within the brain’s olfactory bulb, thereby illuminating potential pathways for sensory information transmission from the periphery to the central nervous system.
“While the olfactory map stands as a significant discovery in its own right, it also furnishes essential groundwork that could empower scientists in the formulation of therapeutic interventions for anosmia, a condition for which effective treatments are presently scarce,” stated Professor Datta.
“An effective resolution to olfactory impairments is contingent upon a fundamental comprehension of its underlying operational principles.”
“For a considerable duration, established cartographies have delineated the organizational principles of receptors within the sensory systems of the eye, ear, and skin, facilitating the capture and interpretation of visual, auditory, and tactile stimuli. Moreover, scientific inquiry has successfully correlated these peripheral maps with their corresponding neural representations within the brain.”
“However, the domain of olfaction has remained an anomaly; it is the sensory modality that has, for the most extended period, lacked a comprehensive spatial representation.”
In their recently concluded study, the investigative team integrated advanced methodologies, including single-cell sequencing and spatial transcriptomics, to meticulously analyze approximately 5.5 million neurons across more than 300 individual murine subjects.
The former technique enabled the precise identification of olfactory receptors expressed by individual neurons within the nasal epithelium, while the latter provided the spatial localization data for these identified receptors.
“This undertaking has resulted in what is arguably the most extensively sequenced neural tissue to date, yet such a magnitude of data was imperative for deciphering the intricacies of this complex system,” Professor Datta remarked.
The researchers discerned that the neuronal population is structured into compact, interdigitating, horizontally oriented bands, extending vertically through the nasal cavity, categorized by the specific type of olfactory receptor they express.
This highly organized receptor topography was found to be remarkably consistent across the study cohort and exhibited a striking parallel with the organizational patterns observed in the olfactory maps of the brain, mirroring the correlations previously documented in the visual, auditory, and tactile sensory systems.
Subsequently, the authors delved into the developmental mechanisms underlying the formation of this nasal olfactory map, identifying retinoic acid—a crucial signaling molecule involved in gene expression regulation—as a principal orchestrator.
Their investigations revealed that a spatial gradient of retinoic acid within the developing nose directs individual neurons to selectively express a particular olfactory receptor based on their established positional coordinates.
Judicious augmentation or reduction of retinoic acid levels precipitated a corresponding upward or downward displacement of the receptor map.
“Our findings demonstrate that developmental processes are capable of achieving this remarkable feat of organizing an extensive repertoire of a thousand distinct olfactory receptors into an exceptionally precise and universally consistent map across different individuals,” Professor Datta affirmed.
A publication detailing these groundbreaking discoveries has been disseminated in the esteemed journal Cell.
_____
A spatial code governs olfactory receptor choice and aligns sensory maps in the nose and brain. Cell, published online April 28, 2026; doi: 10.1016/j.cell.2026.03.051