We often underestimate the significance of our vision. However, recent scientific inquiry has unveiled the remarkable evolutionary trajectory our eyes undertook to achieve their current, recognizable form.
It has been a long-established fact that the ocular structures of vertebrates diverge significantly from those of invertebrates, both in their cellular makeup and developmental processes prior to birth. Nonetheless, the underlying reasons and mechanisms behind the genesis of these divergences remained largely unaddressed for an extended period.
Our comprehensive investigation posits that our ocular origins trace back to a worm-like progenitor that navigated the ancient oceans approximately 600 million years ago. This principle extends to all bilaterally symmetrical animals, a classification encompassing organisms whose bodies can be bisected into approximately mirror-image left and right halves.
As an integral component of this research, we meticulously examined 36 principal classifications of extant fauna, thereby encompassing nearly all bilaterally symmetrical species, to ascertain the disposition and function of their eyes and photoreceptive cells.
A discernible pattern began to materialize. We identified that eyes and light-sensitive cells are consistently situated in two distinct locations: paired symmetrically on either side of the facial region, and centrally atop the cranial structure, directly above the brain.
Among the surveyed organisms, the laterally positioned photoreceptors are instrumental in guiding locomotion, while their centrally located counterparts facilitate the differentiation of day from night and the establishment of vertical orientation.
Our conclusions indicate that an ancestral worm-like organism, from which all vertebrate lineages are derived, relinquished its “steering” paired eyes approximately 600 million years ago, coinciding with its adoption of a predominantly sessile existence. By transitioning to a filter-feeding strategy with no imperative for locomotion, the energetically demanding paired ocular structures became redundant and metabolically costly.
Nevertheless, this shift in lifestyle left the light-sensing cells situated in the dorsal aspect of its head unaffected, as the organism retained the necessity for temporal sensing and orientation. While the paired eyes were lost, the midline photoreceptive cells subsequently underwent development into a rudimentary single eye.
Potentially within several million years, this organism underwent another evolutionary transition. A return to a pelagic existence re-established the requirement for precise steering and the assessment of body movement for efficient filter-feeding (the extraction of sustenance from water) and predator evasion.
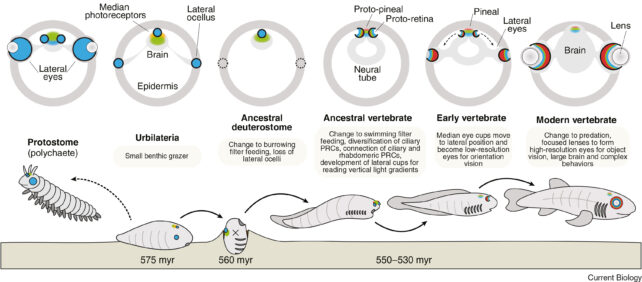
This evolutionary pressure prompted the refinement of the midline eye through the formation of small ocular cups on each lateral aspect. These nascent eye cups subsequently diverged from the central eye, migrated laterally along the skull, and ultimately evolved into the paired ocular structures we possess today: our eyes.
The cessation and subsequent re-establishment of visual capability occurred between 600 and 540 million years ago. Vestiges of the primitive midline eye persisted, differentiating into the pineal organ within the brain, an endocrine structure responsible for the synthesis and release of melatonin, the sleep-regulating hormone.
In a significant proportion of vertebrate species, the pineal organ detects ambient light via a translucent (non-pigmented) region located centrally on the cranium.
However, within the mammalian evolutionary lineage, the pineal organ appears to have forfeited its light-sensing capabilities. This adaptation may be attributed to the nocturnal activity patterns of early mammals, which sought refuge during daylight hours. Consequently, the more photosensitive eyes assumed the primary role of light detection, thereby modulating melatonin secretion and influencing sleep cycles.
A Spectrum of Ocular Morphology
Organisms that retained ancestral paired photoreceptive cells, rather than losing them, constitute the majority of modern invertebrates. These species originated from an evolutionary branch that never adopted a sedentary lifestyle.
This category encompasses a diverse array of creatures, including crustaceans, insects, arachnids, cephalopods, gastropods, and numerous vermiform phyla. These organisms continue to exhibit contemporary iterations of the original light-sensing cellular configurations.
The paired eyes found in insects and crustaceans are characterized as compound eyes, featuring an intricate assembly of minute, densely packed lenses within each ocular unit. In contrast, octopuses and snails possess camera-type eyes, each equipped with a solitary lens.
Remarkably, octopuses and snails have independently achieved comparable ocular designs and demonstrated equivalent visual acuity to that of vertebrates.
Nevertheless, our retinal layer—the light-sensitive tissue lining the posterior of the eye—comprises over 100 distinct neuronal subtypes (with mice exhibiting an even greater number, 140), a stark contrast to the limited quantity found in octopuses and snails. This complexity renders our retina almost as intricate as our cerebral cortex, the expansive outermost layer of the brain.
Prior scientific consensus posited that this level of complexity in our ocular evolution emerged relatively late. Earlier theoretical frameworks, informed by the parallels between brain photoreceptor cells and paired eyes, suggested a rudimentary, pineal organ-like precursor in the early stages of ocular development. However, our current research challenges this notion, contending that a substantial portion of this intricate structure predates the retina itself.
Consequently, it is highly probable that this complexity was already present in the putative “cyclops” ancestor eye. This finding holds significant implications for the genesis and interconnectivity of neural circuits within both our retinas and brains.
For vertebrates, the evolutionary development of our eyes and brains is inextricably intertwined. The emergence of novel paired eyes represents a pivotal element of this narrative, as these structures enabled the sophisticated behaviors that necessitate advanced cognition and substantial cerebral capacity.
Without the evolutionary advent of our eyes, not only would we be bereft of sight; our very existence, along with that of all other vertebrates, would be precluded.






