Emerging scientific findings indicate that specific saccharides originating from intestinal microbiota may serve as an overlooked factor in the pathogenesis of two particularly severe neurodegenerative conditions affecting the brain.
Amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) represent distinct yet interconnected disorders, occasionally exhibiting symptom overlap, both characterized by neuronal demise.
This neuronal loss manifests as impaired muscular control in ALS, which is ultimately fatal, and precipitates profound alterations in behavioral patterns and linguistic faculties in FTD, though motor function can sometimes be compromised in the latter as well.
The precise etiology of these diseases remains elusive. Consequently, an investigative cohort, spearheaded by researchers affiliated with Case Western Reserve University in the United States, sought to ascertain whether the gut microbiome could potentially exert influence, building upon prior evidence suggesting an association with ALS.
Utilizing an experimental murine model, the inquiry identified a particular form of glycogen synthesized by gut microorganisms. Evidence suggests that this carbohydrate instigates cerebral inflammation and precipitates neuronal apoptosis.
“We elucidated that detrimental enteric bacteria generate pro-inflammatory variants of glycogen – a class of sugar – and that these microbial carbohydrates provoke immunological reactions that inflict damage upon the brain,” stated Aaron Burberry, an assistant professor of pathology at Case Western Reserve University.
With this understanding established, therapeutic interventions targeting this pathway can now be pursued.
Both ALS and FTD can be attributed to a specific genomic anomaly in the C9ORF72 gene; however, not all individuals possessing this genetic predisposition develop these conditions.
A primary motivation underpinning this investigation was to delineate other potential triggering agents that may contribute to disease onset among those carrying the genetic variant.
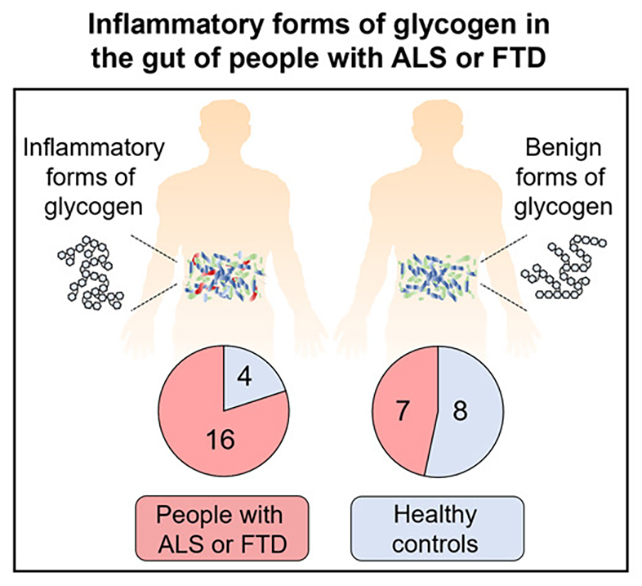
The research team employed a rigorous methodological framework. They generated genetically modified mice lacking the C9ORF72 gene to replicate the human variant and subsequently administered diverse gut bacterial consortia to assess immune system responses.
This experimental approach led to the identification of glycogen production, specifically by Parabacteroides merdae bacteria, one of the identified strains responsible for its synthesis. When introduced into mice devoid of gut flora, this bacterium induced severe inflammation and compromised the integrity of the blood-brain barrier.
Subsequent analyses of human fecal specimens revealed elevated levels of inflammatory glycogen in a significant proportion of ALS patients (15 out of 22) and one FTD patient, contrasting with lower levels observed in only four of twelve healthy control subjects.
“Our findings, demonstrating an enrichment of microbes producing pro-inflammatory glycogen in the intestinal tract of ALS patients, suggest that microbial glycogen may represent a key example among numerous environmental and lifestyle factors that interact with susceptible genotypes to elevate the risk of ALS onset and progression,” observed the investigators in their peer-reviewed publication.
A particularly encouraging outcome of this research was the observation that administering an enzyme known as alpha-amylase, which facilitates glycogen breakdown, to affected mice resulted in attenuated inflammation and prolonged lifespans. While motor function did not show improvement, this finding holds significant therapeutic promise.
This suggests the potential for developing therapeutic strategies that could disrupt this inflammatory cascade, targeting the gut environment rather than the brain directly. However, this aspect of the research is still in its nascent stages.
Scientific understanding continues to expand regarding the intricate connections between the human gut and brain, particularly concerning overall health, and this recent study contributes substantial new insights to this field.
The next phase of the investigation will involve transitioning beyond murine models to conduct more in-depth analyses of human participants and explore various types of glycogen-producing bacteria.
“To elucidate the circumstances and mechanisms leading to the production of detrimental microbial glycogen, the team will proceed with extensive studies examining gut microbiome compositions in ALS/FTD patients both prior to and following disease manifestation,” indicated Burberry.
“Furthermore, our findings provide a foundation for initiating clinical trials to assess whether inhibiting glycogen degradation in ALS/FTD patients could decelerate disease advancement, with such trials potentially commencing within the next year.”