A novel nasal spray developed by researchers has demonstrated the capacity to alleviate cerebral inflammation and reverse memory deficits in laboratory mice.
The scientific cadre at Texas A&M University posits that analogous outcomes may be achievable in human subjects, contingent upon subsequent investigative efforts and therapeutic refinement.
This spray is engineered to address “neuroinflammaging,” characterized by diminutive loci of persistent stress within the aging brain. This condition is strongly correlated with age-associated cognitive deterioration and is implicated in the pathogenesis of neurological ailments like Alzheimer’s disease. The investigators draw a parallel between neuroinflammaging and the continuous operational strain experienced by certain engine components.
Within the experimental murine models, the spray precisely targeted these hyperactive zones by deploying millions of microscopic biological vesicles, recognized as extracellular vesicles (EVs). These EVs, fabricated from human stem cells, are imbued with proteins and genetic directives.
The experimental subjects were 18 months of age, a developmental stage conventionally designated as advanced adulthood in rodent studies, which approximates human individuals in their late fifties to late sixties.
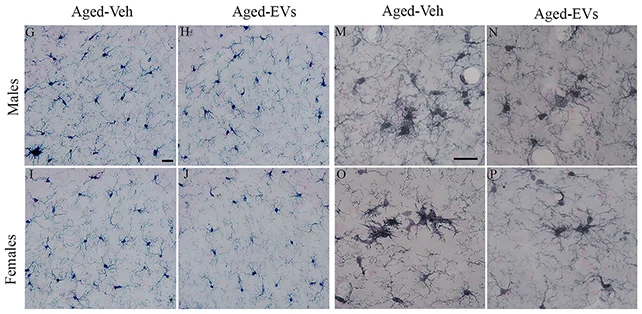
“As we advance the development and broad application of this therapeutic intervention, a straightforward, two-stage nasal administration could potentially supplant invasive, high-risk procedures or even prolonged pharmaceutical regimens,” remarked neuroscientist Ashok Shetty.
“Its applicability is universal. The efficacy of the treatment was consistent and comparable demonstrated across both sexes.”
The research objective was to specifically target microglia, the brain’s resident immune cells, within the hippocampus. This region is pivotal for memory formation and learning and is a site of considerable neuroinflammatory activity.
The utilization of pluripotent stem cells, which possess the capability for differentiation into various cell lineages, represents a therapeutic avenue currently undergoing extensive investigation. It has been empirically established that byproducts of stem cell activity, such as EVs, can exert significant beneficial effects while posing a reduced risk upon systemic administration.
In the context of this investigation, 18-month-old male and female mice were administered two intranasal dosages, separated by a two-week interval.
The nasal delivery route is hypothesized to facilitate direct access of EVs to the brain, circumventing the necessity for surgical intervention, and the researchers have reported rapid assimilation of the vesicles.
Following the dual administration, the treated cohort exhibited superior performance compared to the control group in assessments designed to evaluate object recognition and spatial memory. Subsequent biochemical analyses of hippocampal tissue provided corroboration of the treatment’s intended impact on cerebral inflammation.
Encapsulated within the EVs are microRNAs, minute segments of genetic material instrumental in regulating gene expression. Upon release, these microRNAs facilitate the recalibration of neural circuits, thereby attenuating or silencing the pro-inflammatory signaling pathways implicated in neuroinflammaging.
In aging mice, this mechanism enabled microglia to revert to a more normative functional state, including improvements in mitochondrial energy management.
“MicroRNAs function as principal modulators, orchestrating and governing numerous genetic and signaling cascades within the brain,” explained neuroscientist Madhu Leelavathi Narayana.
“We are effectively restoring neural vitality by mitigating oxidative stress and reinvigorating the brain’s mitochondrial capacity.”
Projections indicate that new instances of dementia in the United States alone could ascend to one million annually by the year 2060, effectively doubling the present incidence. This research offers a potential novel strategy for diminishing age-related neuroinflammation, a recognized trigger for dementia.
Previous studies involving other animal models have suggested that comparable EV-based therapies may also expedite recovery following cerebrovascular accidents or traumatic brain injuries.
While human clinical trials are requisite to ascertain their efficacy in managing conditions such as mild cognitive impairment, the preliminary findings are highly encouraging.
These discoveries resonate with an expanding corpus of scientific inquiry focused on promoting healthier aging, characterized by a reduction in the biological deterioration typically associated with advancing years.
“Neurological disorders associated with aging, such as dementia, represent a paramount global health concern,” stated Shetty.
“Our objective is to foster successful brain aging, ensuring individuals remain engaged, mentally acute, and socially connected. This entails not merely extending lifespan but enhancing the quality of life through improved cognitive function and overall well-being.”